- Search
| Ann Rehabil Med > Volume 44(1); 2020 > Article |
|
Abstract
Objective
To evaluate the prevalence rate, types, characteristics, and associated factors of esophageal dysphagia detected on chest X-ray images after videofluoroscopic swallowing study (VFSS).
Methods
The medical records of 535 adults were reviewed retrospectively. Chest X-ray images taken after barium swallow study were analyzed and presence of any residual barium in the esophagus was considered as esophageal dysphagia. Esophageal dysphagia was classified based on the largest width of barium deposit (mild, <2 cm; severe ≥2 cm) and the anatomic level at which it was located (upper and lower esophagus).
Results
Esophageal residual barium on chest X-ray images was identified in 40 patients (7.5%, 40/535). Esophageal dysphagia was more frequent in individuals aged 65–79 years (odds ratio=4.78, p<0.05) than in those aged <65 years. Mild esophageal dysphagia was more frequent (n=32) than its severe form (n=8). Lower esophageal dysphagia was more frequent (n=31) than upper esophageal dysphagia (n=9). Esophageal residual barium in patients diagnosed with esophageal cancer or lung cancer was significantly associated with severe esophageal dysphagia (p<0.05) and at the upper esophagus level (p<0.01).
Conclusion
Esophageal residual barium was observed on chest X-ray imaging after VFSS. Esophageal barium in the upper esophagus with a diameter of ≥2 cm is an important indicator of malignancy, and chest X-ray image taken after VFSS is an important step to evaluate the presence of esophageal disorder.
The process of swallowing involves the transfer of a food bolus from the oral cavity to the stomach through the oropharynx and esophagus. Disturbed passage of the food bolus can be defined as dysphagia [1]. Esophageal dysphagia can be caused by primary or secondary motility disorders and obstructive lesions. Primary motility disorders include achalasia, nutcracker esophagus, and diffuse esophageal spasm, and secondary motility disorders include scleroderma, muscular dystrophy, myopathy, and amyloidosis. Esophageal carcinoma, lymph node enlargement, or direct invasion can cause obstructive esophageal dysphagia [2].
Oropharyngeal dysfunction has been reported in patients with esophageal dysphagia [3]. Pharyngeal abnormalities were reported in patients with gastroesophageal reflux [4], achalasia [5], and esophageal cancer [6]. Moreover, throat discomfort is a common complaint in patients with esophageal dysphagia; 58% of patients with bolus stuck at the lower esophageal sphincter direct around their neck area [7]. Therefore, a significant portion of patients with a primary clinical diagnosis of oropharyngeal dysphagia could have esophageal dysphagia.
Videofluoroscopic swallowing study (VFSS) is a widely used technique to evaluate oropharyngeal swallowing function and includes a fluoroscopic barium swallow. A chest X-ray image is taken after the barium swallow to detect the presence of barium within bronchus, indicating deep endotracheal aspiration [8]. Normally most of the swallowed barium is observed as gastric content on chest X-ray imaging, contrasting the gastric walls. The esophagus collapses approximately 1 minute after barium swallow in healthy subjects [9]. Therefore, we can consider any esophageal residual barium on chest X-ray imaging as esophageal dysphagia caused by neuromuscular dysmotility or obstructive lesions of esophagus [10].
Most of the previous research that used VFSS as an assessment tool focused on oropharyngeal abnormality. There has been little description of esophageal dysphagia assessed by VFSS. Thus, the purpose of this study was to evaluate the prevalence rate of esophageal dysphagia by the presence of esophageal residual barium on chest Xray imaging after VFSS, and to identify the characteristics and factors associated with esophageal dysphagia.
The medical records of 535 patients aged 19 years or older who underwent VFSS from March 2016 to May 2018 were reviewed retrospectively. All participants were referred for VFSS to evaluate oropharyngeal swallowing dysfunction with various symptoms such as swallowing difficulty of bolus, coughing after swallowing, regurgitation, feeling of food getting stuck, or poor oral intake. Referral diseases were categorized as follows: brain lesions (including stroke, traumatic brain injury, brain tumor, meningitis, and metabolic encephalopathies), dementia, parkinsonism, cancer (including all tumors except brain tumors), other disorders, and unknown cause (i.e., patients who complained of swallowing difficulty without a known underlying cause). Sex, age, and presence of aspiration/penetration during VFSS were also reviewed. Patients were divided into three age groups as <65 years, 65–79 years, and 80 years or older.
VFSS was performed with the patient in an upright seated position. Patients swallowed barium-impregnated boluses of different volumes and consistencies: thin liquid (2 mL and 5 mL), yogurt, pudding, rice gruel, and rice. For the thin fluid test, 43% weight/volume barium sulfate fluid was prepared (Solotop Solution 130; Taejoon Pharmaceutical, Seoul, Korea). Subglottic aspiration indicates that swallowed food material has entered the airway below the vocal folds. In supraglottic penetration, swallowed food material enters the airway, but does not pass the vocal cords. Chest X-ray images were taken approximately 5 minutes after the VFSS (Fig. 1).
Chest X-ray images were analyzed to determine any residual barium in esophagus. We considered any esophageal residual barium on chest X-ray imaging as esophageal dysphagia. In absence of esophageal residual barium on chest X-ray imaging, we classified patients as no esophageal residual barium. Two rehabilitation medicine doctors reviewed the anatomic level of residual esophageal barium and its consequential dilatation severity.
The bronchoaortic constriction is located between pharyngoesophageal and diaphragmatic constrictions. We assessed the locations of tracheal bifurcation and distal end of barium residue on the chest X-ray images (Fig. 2). We classified esophageal dysphagia by the level of the distal end of the residual barium in esophagus as follows: “upper”, cricoid to tracheal bifurcation, and “lower”, tracheal bifurcation to junction of stomach.
After classifying and subgrouping of esophageal dysphagia by severity and level, we compared and analyzed the characteristics of esophageal dysphagia patients with respect to sex, age, referral disease, and presence of aspiration/penetration during VFSS.
In patients with esophageal residual barium, diagnostic studies such as chest computed tomography (CT), gastrofibroscopy, positron emission tomography-computed tomography (PET-CT), electrodiagnostic study, muscle biopsy, and esophagography were also reviewed. Association between causes of esophageal dysphagia and types of esophageal residual barium were analyzed. A case of newly diagnosed cancer-related esophageal dysphagia was reviewed through further medical records.
Statistical analyses were performed using SPSS version 20.0 (IBM, Armonk, NY, USA). Statistical significance was defined as a p-value of <0.05. To investigate factors associated with or without the presence of esophageal residual barium and subgroups according to the severities and levels, the Pearson chi-square test and the likelihood ratio were used. Odds ratios (ORs) were determined using logistic regression analysis to investigate the associated factors of esophageal dysphagia. The association between severity and level of esophageal dysphagia was analyzed by the Pearson chi-square test. The Fisher exact test was used to analyze the association between the causes and the determined subgroups of esophageal dysphagia.
The mean age of the 535 participants was 73.5 years, and 60.7% were men. Forty patients (7.5%) had esophageal barium residue present on their chest X-ray images and were defined as having esophageal dysphagia.
The mean age of patients with esophageal dysphagia (n=40) was 75.3±9.2 years, and 24 (60%) were men. The most common age group was 65–79 years (n=28, 70.0%), followed by 80 years or older and <65 years. The most common referral disease was brain lesions (n=20, 50.0%), followed by unknown causes, dementia, cancer, and other disorders. Other disorders included vocal cord palsy, chronic obstructive pulmonary disease, and cervical spine fusion operation. No participants had parkinsonism. The age group was significantly different between patients with esophageal dysphagia and no esophageal residual barium (p=0.002). Sex, referral disease, and aspiration/penetration during VFSS were not significantly associated with the presence of esophageal residual barium (Table 1).
Characteristics with p-value less than 1.0 were analyzed for their association with esophageal dysphagia. Age group and referral disease were selected for the univariable logistic regression analysis. The 65–79 years age group had more patients with esophageal dysphagia (OR=4.78; 95% confidence interval [CI], 1.42–16.07; p<0.05) compared to the <65 years age group, and the ≥80 years age group (p=0.37). Patients with unknown referral disease significantly exhibited esophageal dysphagia (OR=2.42; 95% CI, 1.13–5.16; p<0.05) compared to patients with brain lesions (Table 2).
In esophageal dysphagia patients, mild (<2 cm) esophageal dysphagia was detected in 32 (80.0%) patients, and severe (≥2 cm) esophageal dysphagia in 8 (20.0%) patients. Lower esophageal barium (n=31, 81.3%) was more frequent than upper esophageal barium (n=9, 22.5%). The severity and level of esophageal dysphagia were not significantly related, as per chi-square analysis (Pearson χ=1.29; p=0.26).
Characteristics such as sex, age, referral disease, and aspiration/penetration during VFSS did not show significant relationship with severity and level of esophageal dysphagia. Regarding the referral diseases, dementia was frequent in lower level while cancer was more frequent in upper and severe subgroups, but without statistical significance (p=0.06 for levels; p=0.10 for severities) (Table 3).
Among the 40 patients with esophageal residual barium, 15 patients underwent evaluations for esophageal abnormality: gastrofibroscopy (n=12), chest CT (n=9), PET-CT (n=2), electrodiagnostic study (n=2), muscle biopsy (n=1), and esophagography (n=1). Esophagitis, myopathy, achalasia, and cancer were identified in 10 patients. No distinct causes of esophageal dysphagia were identified in 5 patients (Table 4).
The causes of esophageal dysphagia in these 15 patients were compared with esophageal anatomic levels (upper and lower esophagus) and severities (mild and severe). Esophagitis, myopathy, achalasia, and esophageal dysphagia with unidentified cause were not significantly associated with the level or severity of esophageal dysphagia. Cancer-related esophageal dysphagia showed significant association with upper esophageal dysphagia (p<0.01) and severe esophageal dysphagia (p<0.05), based on the Fisher exact test (Table 5). Of the three patients with cancer-related esophageal dysphagia, one was newly diagnosed after VFSS. Severe esophageal dilatation was observed at upper esophagus level on chest X-ray images after VFSS; and subsequent chest CT identified a subcarinal mass, confirmed as non-small cell lung cancer (Fig. 3).
In this study, we assessed esophageal dysphagia by identifying residual barium in the esophagus on chest X-ray images taken after VFSS. Esophageal dysphagia is characterized as an impedance in the passage of food through esophagus due to structural abnormality or dysmotility [11]. Compared to oropharyngeal dysphagia, there are limited data on the prevalence or incidence of esophageal dysphagia. There are few published articles assessing esophageal abnormalities on chest X-ray images after VFSS, such as delayed esophageal transit time or the presence of esophageal stricture, obstruction, and diverticulum [12,13].
Among 535 patients with dysphagia, the prevalence of esophageal dysphagia was 7.5% (40/535). The occurrence of penetration and aspiration was not significantly different between patients with and without esophageal residual barium. Among the esophageal dysphagia patients, 57.5% exhibited penetration and 20.0% exhibited aspiration, while 42.0% and 27.7% of the patients without esophageal residual barium exhibited penetration and aspiration, respectively. In a previously published report, the presence of aspiration and esophageal abnormality were also not significantly associated [12], suggesting that the status of esophageal swallowing needs to be assessed regardless of the presence of oropharyngeal dysphagia.
We defined esophageal dysphagia as the presence of any residual barium in the esophagus identified on chest X-ray imaging. If there is no esophageal residual barium on chest X-ray imaging, we classified patients as no esophageal residual barium. In a timed barium esophagogram study for achalasia, patients with normal esophageal function exhibited a maximum esophageal width of 2 cm on chest X-ray images after ingesting a barium contrast agent, and the esophagus collapsed after approximately 1 minute [9]. In a similar study, healthy volunteers exhibited a maximum esophageal width of <1 cm on chest X-ray images taken 1 minute after swallowing barium [14]. In the current study, chest X-ray images were taken approximately 5 minutes after VFSS, and thus presence of residual barium in esophagus can be considered as abnormal esophageal motility. The normal anterior-posterior dimension of the esophagus is 1–2 cm [15], and when the largest width of esophageal barium exceeded 2 cm, we considered it as severe esophageal dysphagia.
Anatomically, there are three major constriction points in esophagus, which are natural esophageal narrowing [16–18]. The pharyngoesophageal constriction is at the junction of the pharynx and esophagus. The bronchoaortic constriction is located behind the tracheal bifurcation and at the crossing of the aortic arch and the left main bronchus. The third, diaphragmatic constriction, is located at the junction with the stomach. Relative to the bronchoaortic constriction point, the esophagus can be defined as the upper and lower esophagus [18]. We used the bronchoaortic constriction as the reference level of esophageal dysphagia.
We aimed to evaluate the factors associated with esophageal dysphagia. Age group of 65–79 years was associated with moderate to severe esophageal dilatation and showed a significant relationship with esophageal dysphagia. Reduced esophageal motility in old age has been attributed to weak peristalsis, slow bolus progression, and decreased esophageal sphincter pressure [19–21]. In the current study, based on the presence of esophageal residual barium after VFSS, older age is associated with esophageal dysphagia, a finding consistent with previous reports.
In our analysis, age over 80 years was not significantly associated with esophageal dysphagia. Very few studies have focused on the relationship between the more elderly age group and esophageal motility [20–22]. Andrews et al. [23] compared esophageal motility of age over 80 years with a younger age group (mean age, 35 years), and there was no significant difference in the functional assessment and manometric findings. A large-scale research would be necessary to investigate the relationship between esophageal motility and a more elderly age group.
In our study, lower esophageal barium residue (n=31, 81.3%) and severe esophageal dilatation (n=32, 80.0%) were more frequently observed. Of the causal diseases of esophageal dysphagia, cancer related dysphagia was more frequent in upper level, with severe dilatation.
Many esophageal dysphagia cases caused by intrinsic or extrinsic obstructive lesions are due to malignancy [2]. Esophageal cancer is the seventh most common cancer and the sixth most common cause of cancer-related death worldwide [24]. Moreover, the prevalence of esophageal cancer is increasing [25]. The most common histological types of esophageal cancer are squamous cell carcinomas and adenocarcinomas. Squamous cell carcinomas are more commonly distributed in upper and middle thirds of esophagus; while adenocarcinomas are more common in distal esophagus and associated with gastroesophageal reflux disorder and Barrett’s esophagus [26,27]. Shin et al. [28] reported that, in Korea, squamous cell carcinoma was the most common histological type (90.2%) of all esophageal cancer in 2013, followed by adenocarcinoma (3.1%). In our study, two patients had esophageal cancer with severe dilatation in upper esophagus as seen on chest X-ray imaging, histologically confirmed as squamous cell carcinoma. Our results correspond to the high prevalence of squamous cell carcinoma in Korea and the anatomic level of esophageal cancer.
Lung cancer is also associated with esophageal dysphagia, with four possible mechanisms: extrinsic compression of the esophagus within the mediastinum, compression of the pharynx and upper esophagus, esophageal stenosis secondary to radiotherapy, and secondary achalasia [29,30]. In our study, one patient showed severe upper esophageal dilatation on chest X-ray imaging, and was newly diagnosed as non-small cell lung cancer after VFSS.
Barium esophagography, esophageal scintigraphy, manometry, and endoscopic evaluation are all techniques for evaluating esophageal abnormalities. Scintigraphy or manometry are not widely used, and endoscopy is not applicable for functional assessment of the esophagus [31]. Barium esophagography is a commonly used technique because it is noninvasive, inexpensive, and easily available for evaluating the pharynx and esophagus [10]. In one study, in patients with achalasia or those treated with pneumatic dilatation, timed barium esophagogram was performed to study the esophageal emptying time. This was assessed by measuring the height and width or the area of the barium column on images taken at 1, 2, and 5 minutes after barium ingestion. Barium empties from the esophagus in 1 minute in most and in 5 minutes in all healthy individuals [31].
Thus, barium esophagography is recommended to assess esophageal dysfunction, while oropharyngeal-swallowing function is not assessed in order to avoid massive aspiration of barium in patients with oropharyngeal dysphagia. Although barium sulfate is not toxic, massive amounts of barium could result in pulmonary complications like pneumonitis [32,33]. We recommend VFSS as initial assessment for patients with, or who are suspected to have, oropharyngeal dysphagia.
There are several limitations to this study. This was a single-institution based retrospective study; thus, our patients may not represent the characteristics of the general population, and a confounder bias could exist. There is also a potential risk of selection bias due to the subgroup (n=15) analysis who underwent workup evaluations among the 40 patients with esophageal dysphagia. The results of esophageal dysphagia evaluation were not validated by comparing them to a gold standard. Few of the patients with esophageal dysphagia had been examined through diagnostic work-up using chest CT or endoscopic evaluation, but not manometry. Manometry can provide important information regarding esophageal motility and esophageal pressure in patients who are suspected of having esophageal dysmotility disorders such as achalasia; however, manometry was not available in our institution during the study period. Further evaluation of esophageal dysphagia by VFSS with manometry is necessary. Finally, the time interval between barium swallowing and chest X-ray imaging was not strictly controlled. In all patients, chest X-ray images were taken as soon as possible after barium swallowing, approximately after 5 minutes. Some patients who showed no esophageal residual barium in chest X-ray imaging might have mild esophageal dysphagia. Despite of these limitations, this is the first study to assess esophageal dysphagia using chest X-ray imaging following VFSS and to evaluate the prevalence and type of esophageal dysphagia, as well as to identify its characteristics and associated factors. Future large-scale studies on esophageal dysphagia that consider alternative diagnostic techniques are required to validate and extend our findings.
In conclusion, to our knowledge, there are no studies evaluating the characteristics and associated factors of esophageal dysphagia using abnormal esophageal findings identified on X-ray images in VFSS. We recommend VFSS as an initial assessment for patients with, or who are suspected to have oropharyngeal dysphagia. Here, the prevalence of esophageal dysphagia was 7.5% of the study population, as identified by chest X-ray imaging following VFSS, and esophageal dysphagia was frequent in patients with and without oropharyngeal dysphagia. Not all cases of esophageal dysphagia with proximal severe dilatation are indicative of malignancies; thus, chest X-ray images taken after VFSS are an important step to evaluate the presence of the esophageal disorder.
AUTHOR CONTRIBUTION
Conceptualization: Min YK, Baek S. Methodology: Baek S. Visualization: Min YK, Baek S. Writing – original draft: Min YK, Baek S. Writing – review and editing: Baek S, Kang EK, Nam SJ. Approval of final manuscript: all authors.
Fig. 1.
Flow diagram of esophageal residual barium in chest X-ray taken immediately after videofluoroscopic swallowing study (VFSS).
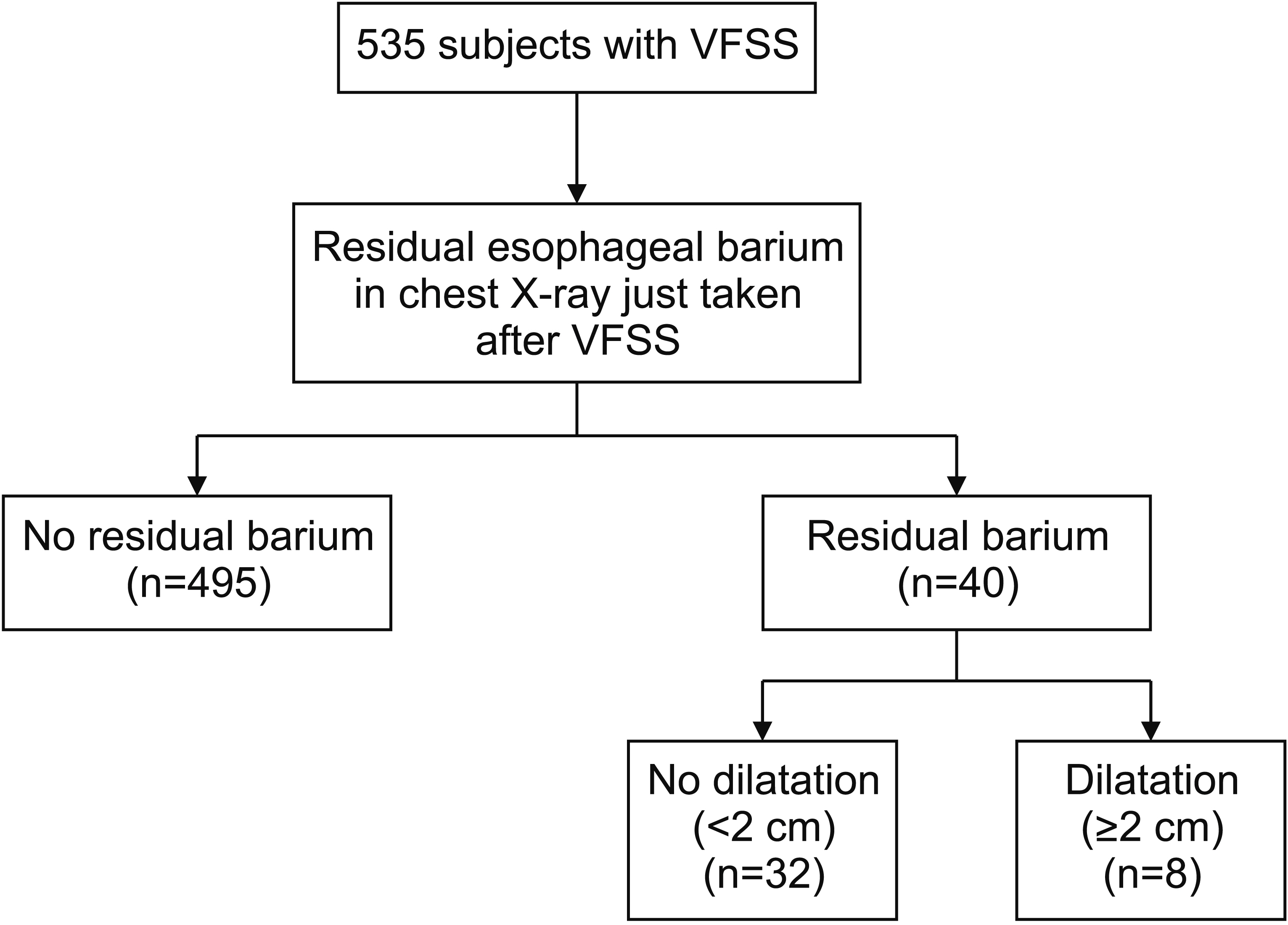
Fig. 2.
Severity and level of esophageal dysphagia. Largest diameter (arrow) of 15.85 mm was classified as mild esophageal dysphagia. The distal end of the barium residue (asterisk) proximal to the tracheal bifurcation was classified as upper esophageal dysphagia. The distal trachea and bifurcation are delineated as black solid line.
Fig. 3.
Imaging studies of the case who newly diagnosed as a cancer-related esophageal dysphagia after videofluoroscopic swallowing study (VFSS). The patient presented for unexplained swallowing difficulty with suffering from poor oral intake. (A) After VFSS, severe esophageal dilatation is observed at upper esophagus level (arrowhead) in the chest X-ray. (B) The esophagus, with a nasogastric tube, is compressed by the subcarinal mass (asterisk), and (C) hypermetabolism is seen on the positron emission tomography-computed tomography scan.
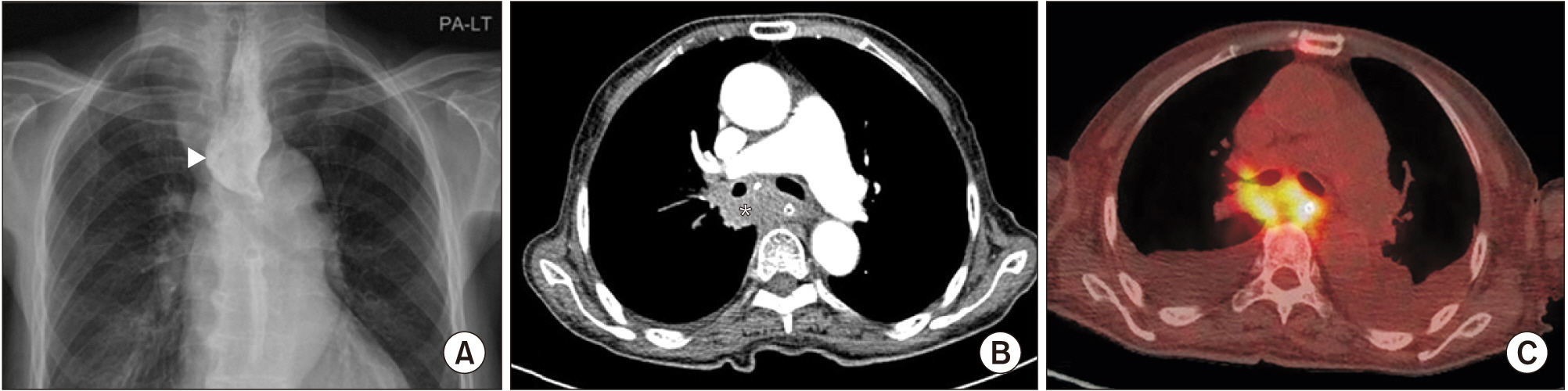
Table 1.
Characteristics of patients with esophageal dysphagia (ED) or no esophageal residual barium (no residue)
| All (n=535) | No residue (n=495) | ED (n=40) | p-value | |
|---|---|---|---|---|
| Sex | 0.92a) | |||
| Male | 325 (60.7) | 301 (60.8) | 24 (60.0) | |
| Female | 210 (39.3) | 194 (39.2) | 16 (40.0) | |
| Age (yr) | 0.002*b) | |||
| <65 | 111 (20.7) | 108 (21.8) | 3 (7.5) | |
| 65–79 | 239 (44.7) | 211 (42.6) | 28 (70.0) | |
| ≥80 | 185 (34.6) | 176 (35.6) | 9 (22.5) | |
| Referral disease | 0.09b) | |||
| Brain | 322 (60.2) | 302 (61.0) | 20 (50.0) | |
| Dementia | 50 (9.3) | 46 (9.3) | 4 (10.0) | |
| Parkinsonism | 30 (5.6) | 30 (6.1) | - | |
| Cancer | 13 (2.4) | 11 (2.2) | 2 (5.0) | |
| Unknown | 87 (16.3) | 75 (15.2) | 12 (30.0) | |
| Others | 33 (6.2) | 31 (6.3) | 2 (5.0) | |
| VFSS findings | 0.17b) | |||
| No A/P | 157 (29.3) | 150 (30.3) | 9 (22.5) | |
| P | 231 (43.2) | 208 (42.0) | 23 (57.5) | |
| A | 147 (27.5) | 137 (27.7) | 8 (20.0) |
Table 2.
Factors associated with esophageal dysphagia compared to no esophageal residual barium using univariate logistic regression analyses
| OR (95% CI) | p-value | |
|---|---|---|
| Age (yr) | ||
| <65 | 1 | |
| 65–79 | 4.78 (1.42–16.07) | 0.01* |
| ≥80 | 1.84 (0.49–6.95) | 0.37 |
| Referral disease | ||
| Stroke | 1 | |
| Dementia | 1.31 (0.43–4.01) | 0.63 |
| Parkinsonism | - | 1.00 |
| Cancer | 2.75 (0.57–13.24) | 0.21 |
| Unknown | 2.42 (1.13–5.16) | 0.02* |
| Others | 0.97 (0.22–4.37) | 0.97 |
Table 3.
Characteristics according to subgroups of esophageal dysphagia
|
Level |
p-value |
Severity |
p-value | |||
|---|---|---|---|---|---|---|
| Upper (n=9) | Lower (n=31) | Mild (n=32) | Severe (n=8) | |||
| Sex | 0.06 | 0.92a) | ||||
| Male | 3 (33.3) | 21 (67.7) | 18 (56.3) | 6 (75.0) | ||
| Female | 6 (66.7) | 10 (32.3) | 14 (43.8) | 2 (25.0) | ||
| Age (yr) | 0.24 | 0.32b) | ||||
| <65 | 0 (0) | 3 (9.7) | 3 (9.4) | 0 (0) | ||
| 65–79 | 8 (88.9) | 20 (64.5) | 21 (65.6) | 7 (87.5) | ||
| ≥80 | 1 (11.1) | 8 (25.8) | 8 (25.0) | 1 (12.5) | ||
| Referral disease | 0.06 | 0.10b) | ||||
| Brain | 4 (44.4) | 16 (51.6) | 17 (53.1) | 3 (37.5) | ||
| Dementia | 0 (0) | 4 (12.9) | 3 (9.4) | 1 (12.5) | ||
| Cancer | 2 (22.2) | 0 (0) | 0 (0) | 2 (25.0) | ||
| Unknown | 3 (33.3) | 9 (29) | 10 (31.3) | 2 (25.0) | ||
| Others | 0 (0) | 2 (6.5) | 2 (6.3) | 0 (0) | ||
| VFSS findings | 0.25 | 0.83b) | ||||
| No A/P | 3 (33.3) | 6 (19.4) | 7 (21.9) | 2 (25.0) | ||
| P | 3 (33.3) | 20 (64.5) | 18 (56.3) | 5 (62.5) | ||
| A | 3 (33.3) | 5 (16.1) | 7 (21.9) | 1 (12.5) | ||
Table 4.
Evaluations of the causes of esophageal dysphagia in 15 participants
| Case no. | Age (yr) | Sex | Referral disease |
Esophageal dysphagia |
Work-up evaluations for the cause of esophageal dysphagia | Cause of esophageal dysphagia | |
|---|---|---|---|---|---|---|---|
| Level | Severity | ||||||
| 1 | 75 | M | Esophageal cancer | Upper | Severe | Chest CT | Esophageal cancera) |
| 2 | 75 | M | Esophageal cancer | Upper | Severe | Gastrofibroscopy, chest CT, PET-CT | Esophageal cancera) |
| 3 | 77 | F | Unknown cause of swallowing difficulty | Upper | Severe | Gastrofibroscopy, chest CT, PET-CT | Non-small cell lung cancer |
| 4 | 78 | F | Internal capsular infarction | Upper | Mild | Gastrofibroscopy, chest CT | Not significant |
| 5 | 77 | M | Internal capsular infarction | Lower | Severe | Gastrofibroscopy, chest CT, esophagography | Achalasiaa) |
| 6 | 71 | M | Unknown cause of swallowing difficulty | Lower | Severe | Gastrofibroscopy | Candida esophagitisa) |
| 7 | 78 | M | Dementia | Lower | Severe | Gastrofibroscopy | Not significant |
| 8 | 71 | M | Unknown cause of swallowing difficulty | Lower | Mild | Gastrofibroscopy, chest CT, electromyography | Dermatomyositis |
| 9 | 53 | M | Unknown cause of swallowing difficulty | Lower | Mild | Gastrofibroscopy, electromyography, muscle biopsy | Mitochondrial myopathy |
| 10 | 66 | M | Unknown cause of swallowing difficulty | Lower | Mild | Gastrofibroscopy | Candida esophagitis |
| 11 | 75 | M | Pontine infarction | Lower | Mild | Gastrofibroscopy | Erosive esophagitis |
| 12 | 71 | M | Multiple cerebral infarction | Lower | Mild | Gastrofibroscopy | Reflux esophagitis (LA grade B) |
| 13 | 76 | M | Unknown cause of swallowing difficulty | Lower | Mild | Chest CT | Not significant |
| 14 | 68 | F | Capsulothalamic infarction | Lower | Mild | Chest CT | Not significant |
| 15 | 91 | F | Unknown cause of swallowing difficulty | Lower | Mild | Gastrofibroscopy, chest CT | Not significant |
Table 5.
Associations between cause of esophageal dysphagia and type of esophageal dysphagia (n=15)
| Causes of esophageal dysphagia |
Level |
p-value |
Severity |
p-value | ||
|---|---|---|---|---|---|---|
| Upper (n=4) | Lower (n=11) | Mild (n=6) | Severe (n=9) | |||
| Not identified (n=5) | 1 | 4 | 1.00 | 4 | 1 | 0.58 |
| Esophagitis (n=4) | 0 | 4 | 0.52 | 3 | 1 | 0.60 |
| Myopathy (n=2) | 0 | 2 | 1.00 | 2 | 0 | 0.49 |
| Achalasia (n=1) | 0 | 1 | 1.00 | 0 | 1 | 0.40 |
| Cancer (n=3) | 3 | 0 | 0.009** | 0 | 3 | 0.04* |
REFERENCES
3. Triadafilopoulos G, Hallstone A, Nelson-Abbott H, Bedinger K. Oropharyngeal and esophageal interrelationships in patients with nonobstructive dysphagia. Dig Dis Sci 1992;37:551-7.



4. Mendell DA, Logemann JA. A retrospective analysis of the pharyngeal swallow in patients with a clinical diagnosis of GERD compared with normal controls: a pilot study. Dysphagia 2002;17:220-6.



5. Jones B, Donner MW, Rubesin SE, Ravich WJ, Hendrix TR. Pharyngeal findings in 21 patients with achalasia of the esophagus. Dysphagia 1987;2:87-92.



6. Martin RE, Letsos P, Taves DH, Inculet RI, Johnston H, Preiksaitis HG. Oropharyngeal dysphagia in esophageal cancer before and after transhiatal esophagectomy. Dysphagia 2001;16:23-31.



7. Smith DF, Ott DJ, Gelfand DW, Chen MY. Lower esophageal mucosal ring: correlation of referred symptoms with radiographic findings using a marshmallow bolus. AJR Am J Roentgenol 1998;171:1361-5.


8. Han TR, Paik NJ, Park JW. The safety of videofluoroscopic swallowing study (VFSS). J Korean Acad Rehabil Med 2000;24:215-8.
9. Kostic SV, Rice TW, Baker ME, Decamp MM, Murthy SC, Rybicki LA, et al. Timed barium esophagogram: a simple physiologic assessment for achalasia. J Thorac Cardiovasc Surg 2000;120:935-43.


10. Levine MS, Rubesin SE, Laufer I. Barium esophagography: a study for all seasons. Clin Gastroenterol Hepatol 2008;6:11-25.


12. Miles A, McMillan J, Ward K, Allen J. Esophageal visualization as an adjunct to the videofluoroscopic study of swallowing. Otolaryngol Head Neck Surg 2015;152:488-93.


13. Watts S, Gaziano J, Jacobs J, Richter J. Improving the diagnostic capability of the modified barium swallow study through standardization of an esophageal sweep protocol. Dysphagia 2019;34:34-42.



14. Kostic S, Andersson M, Hellstrom M, Lonroth H, Lundell L. Timed barium esophagogram in the assessment of patients with achalasia: reproducibility and observer variation. Dis Esophagus 2005;18:96-103.



15. Kuo B, Urma D. Esophagus-anatomy and development. GI Motil Online 2006 [Epub]. https://www.nature.com/gimo/contents/pt1/full/gimo6.html.
16. Chikwe J, Cooke D, Weiss A. Esophagus. Cardiothoracic surgery. 2nd ed. Oxford, UK: Oxford University Press; 2013.
17. Thorek P. Esophagogastrointestinal tract. In: Anatomy in surgery, New York, NY: Springer; 1985. p.418-513.
18. Skandalakis JE, Skandalakis LJ. Esophagus. In: Skandalakis JE, Skandalakis PN, Skandalakis LJ, editors. Surgical anatomy and technique: a pocket manual. 2nd ed. New York, NY: Springer; 2000.
19. Khan TA, Shragge BW, Crispin JS, Lind JF. Esophageal motility in the elderly. Am J Dig Dis 1977;22:1049-54.



20. Lee J, Anggiansah A, Anggiansah R, Young A, Wong T, Fox M. Effects of age on the gastroesophageal junction, esophageal motility, and reflux disease. Clin Gastroenterol Hepatol 2007;5:1392-8.


21. Shim YK, Kim N, Park YH, Lee JC, Sung J, Choi YJ, et al. Effects of age on esophageal motility: use of highresolution esophageal impedance manometry. J Neurogastroenterol Motil 2017;23:481.




22. Gutschow CA, Leers JM, Schroder W, Prenzel KL, Fuchs H, Bollschweiler E, et al. Effect of aging on esophageal motility in patients with and without GERD. Ger Med Sci 2011;9:Doc22.


23. Andrews JM, Fraser RJ, Heddle R, Hebbard G, Checklin H. Is esophageal dysphagia in the extreme elderly (>or=80 years) different to dysphagia younger adults? A clinical motility service audit. Dis Esophagus 2008;21:656-9.



24. International Agency for Research on Cancer. Cancer fact sheets [Internet]. Lyon, France, International Agency for Research on Cancer. c2018;[cited 2020 Jan 15]. http://gco.iarc.fr/today/fact-sheets-cancers.
26. Napier KJ, Scheerer M, Misra S. Esophageal cancer: a review of epidemiology, pathogenesis, staging workup and treatment modalities. World J Gastrointest Oncol 2014;6:112-20.



28. Shin A, Won YJ, Jung HK, Kong HJ, Jung KW, Oh CM, et al. Trends in incidence and survival of esophageal cancer in Korea: analysis of the Korea Central Cancer Registry Database. J Gastroenterol Hepatol 2018;33:1961-8.



29. Makker HK, Chisholm R, Rate AJ, Bancewicz J, Bernstein A. Dysphagia due to secondary achalasia as an early manifestation of squamous cell carcinoma. Postgrad Med J 1995;71:502-4.



31. Neyaz Z, Gupta M, Ghoshal UC. How to perform and interpret timed barium esophagogram. J Neurogastroenterol Motil 2013;19:251-6.




- TOOLS
-
METRICS

- Related articles in ARM
-
Characteristics of Dysphagia in Children with Cerebral Palsy via Videofluoroscopy.2000 August;24(4)






