- Search
| Ann Rehabil Med > Volume 47(4); 2023 > Article |
|
Abstract
Objective
To determine the effects of lower limb muscle fatigue on spatiotemporal gait parameters and turning difficulty characteristics during the extended Timed Up and Go (extended TUG) test in individuals with different severity stages of Parkinson’s disease (PD).
Methods
Forty individuals with PD, classified as Hoehn and Yahr (H&Y) stages 2 and 3 participated in this pre- and post-experimental study design. The participants performed a continuous sit-to-stand task from a chair based on 30 cycles/min set-up to induce lower limb muscle fatigue. They performed extended TUG test immediately before and after completing the fatigue protocol. Spatiotemporal gait parameters and turning difficulty characteristics were recorded using two GoPro® Hero 4 Silver cameras. Data were subjected to a repeated-measure ANOVA.
Results
Individuals with PD experience significant changes in spatiotemporal gait parameters, specifically stride velocity and length, under conditions of lower limb muscle fatigue (p=0.001). These changes were more pronounced in individuals with PD in the H&Y stage 3 group. Additionally, both PD groups exhibited difficulty with turning, requiring more than five steps to complete a 180° turn and taking more than 3 seconds to accomplish it.
Conclusion
These findings highlight the impact of muscle fatigue on gait performance in PD and suggest that individuals in later stages of the disease may be particularly affected. Further research is needed to explore interventions that can mitigate these gait impairments and improve mobility in individuals with PD.
Turning during walking is a challenging locomotor task due to the need for a complex integration among different motor control mechanisms [1]. Turning task requires the central nervous system to coordinate several body components to help change the direction while preserving the postural stability in the medio-lateral direction and continuing the ongoing step cycle [2]. A turn changes the direction of walking with a top-down coordination that begins with head rotation, followed by the trunk, pelvis, and feet, with body parts moving toward the inner side of the turn [3]. Difficulty in turning is one of the early gait abnormalities in individuals with Parkinson’s disease (PD). These individuals suffer from several gait challenges during straight walking and turning while walking, which are even more challenging in the early stages of the disease [1,4]. As the disease progresses, individuals with PD present with reduced gait speed followed by shuffling gait and shorter steps, diminished arm swings bilaterally, and slower “en-bloc” turns, making it difficult to navigate turns or change in direction [5]. This phenomenon can result in falls due to losing balance when the patient suddenly stops moving or becomes unsteady.
Gait performance is influenced by age, gait speed, depression, fatigue, motor severity, executive function, and attention. These factors can exacerbate motor symptoms like bradykinesia, rigidity, and postural instability, leading to difficulty initiating and maintaining a steady gait [6]. Fatigue can further impair gait function in individuals with PD. Muscle strength and coordination may decrease, leading to increased difficulty initiating and maintaining a steady gait [7]. Furthermore, lower limb muscle fatigue among individuals with PD may limit their ability to pass or cross an obstacle [8]. A new segmental organization is required to maintain motor performance, as indicated by changes in spatiotemporal gait parameters during muscle fatigue [9]. Therefore, individuals with PD may demonstrate further spatiotemporal gait adjustments due to greater muscle fatigue and deterioration in motor control mechanisms [10].
Muscle fatigue among individuals with PD has gained increasing attention. An earlier study demonstrated no clear association between gait and gait-related activities and fatigue in people with PD [11]. By contrast, individuals with PD showed increased stride length and speed, reduced stride duration, and vertical braking impulses [12]. Another study found that lower limb muscle fatigue has contributed to slower walking speed among individuals with PD [13]. However, no significant decline in gait characteristics has been reported following lower limb muscle fatigue [14]. The discrepancies in existing studies may be due to differences in inducing fatigue protocols, outcome measures used, group of muscles measured, and patient characteristics. To date, no study has investigated the effects of lower limb muscle fatigue on spatiotemporal gait parameters during the turning of individual with PD. Therefore, the effects of fatigue conditions on gait parameters should be investigated to gain further insights into gait recovery in PD.
The Timed Up and Go (TUG) test has been widely used as a clinical measure of balance and mobility [15]. This reliable test determines the ability and time needed to perform basic functional mobility tasks. The TUG test compromises fundamental activities of daily living, such as standing up, walking for 3 m, turning around, and sitting back [16-19]. The walking distance increased from 3 m in the classical TUG to 7 m in the extended TUG test to improve the ability to capture balance and gait impairments [20]. The reliability of the extended test was good [21]. The extended TUG test has a good association with gait speed and lower limb strength.
A previous study used the TUG test to capture the characteristics of turning difficulty in the older person [3]. They identified four indicators of turning difficulty: staggering, absence of pivoting, taking five or more steps, and taking 3 seconds or longer to achieve a turn. The characteristics of the TUG test make it a good tool for determining the effects of muscle fatigue conditions on gait parameters during walking and turning among individuals with PD. Exploring the characteristics of turning difficulty due to lower limb muscle fatigue is important to guide the development of a management plan for individuals with PD who are at high risk of falling. Therefore, this study aimed to determine the effects of lower limb muscle fatigue on spatiotemporal gait parameters during the straight walking and turning phases of the extended TUG test in individuals with PD. Another goal was identifying the turning difficulty characteristics after lower limb muscle fatigue. We hypothesized that lower limb muscle fatigue would lead to a greater effect on gait parameters for individuals with PD with higher severity than those with lower severity by using Hoehn and Yahr (H&Y) classification.
Forty individuals with PD were recruited from a selected government-funded hospital with established services for neurological conditions. The effect size was calculated from a previous study [12]. The estimated sample was 40 participants to provide 90% power, with a risk of type 1 error of 0.05. All participants signed an informed consent form approved by the Research Ethics Committee, Universiti Teknologi MARA (REC/44/18) and Medical Research Ethics Committee (MREC), Ministry of Health, Malaysia (NMRR-17-3490-39360 IIR).
The inclusion criteria were as follows: (1) diagnosis of PD with stages 2 or 3 based on H&Y classification confirmed by a neurologist; (2) Mini-Mental State Examination score≥24; (3) ability to walk 5 minutes without assistance or use of any aids; and (4) age between 45 and 80 years at the time of recruitment. Participants were excluded from the study if they presented visual field defects based on confrontation technique, had other diseases or injuries that could potentially disturb gait, or had artificial joints or orthotic devices and unstable co-existing medical conditions.
This study used a modified TUG test, referred to hereafter as an extended TUG test. The extended TUG protocol with 7 m of walking may have been chosen to provide a longer distance for assessing gait performance and capture more gait cycles [22]. Furthermore, the 7 m distance may have been considered sufficient for evaluating functional recovery and mobility in certain populations, such as individuals with PD [23]. The reliability of the extended TUG test in the estimation of turn and turn-to-sit was high (p>0.75). A previous study suggested that the extended TUG test performance is a useful indicator of cognition, motor function, and quality of life among individuals with PD [20].
The participants were instructed not to perform strenuous physical activity for 48 hours before the testing date. All assessments were conducted with patients in the “on” status, with participants taking their medications one hour before the assessment. The assessment procedure was completed within 2 hours to ensure participants were in the “on” status. The experiment was conducted in a hospital-based physiotherapy gymnasium. Participants’ demographic and medical data were collected before testing. After the clinical assessment, the participants were asked to perform the extended TUG test [24].
Before testing, the researcher explained and demonstrated the procedures to the participants. They were required to stand up from a sitting position (without using their arms) and then walk at the marked distance of 7 m in a straight line at their comfortable self-selected speed. At the end of 7 m, the participants were required to make a 180° turn and then return to the starting chair and sit. The tester used a stopwatch to measure the total time needed to complete the extended TUG task. The participants were allowed to carry out a one-time practice trial before the real test, the result of which was not used for analysis. The participants performed walking tasks thrice at their self-selected speed before the lower limb muscle fatigue protocol. The average time to complete the test was taken as the baseline measurement. The participants were allowed to wear their usual footwear. The layout for the extended TUG test was marked on the floor (7 m and turning areas), and these marks were clearly shown to the participants.
The lower limb muscle fatigue protocol was adopted from a previous study, in which the participants performed a continuous sit-to-stand task from a chair with arms across the chest region to induce lower limb muscle fatigue [12]. A standard chair without armrests was used in this task (43 cm in height, 41 cm in width, and 42 cm in depth). A metronome with 30 cycles/min set-up was used to control the frequency of the sit-to-stand movement. The instructions to the participants were as follows: “with the beat of a metronome, please stand up into an upright position and then sit down; and repeat the task until you can no longer perform the task.” Initially, the tester demonstrated to the participants the whole procedure, and the participants were allowed a one-time trial as familiarization. During the fatigue protocol, the assessor verbally encouraged the participants to continue the task until they felt too exhausted to do more repetitions. The participants were stopped from continuing the task when they met one of the following criteria: the task frequency was less than 30 cycles/min after encouragement, after 30 minutes of the sit-to-stand task, or when the participants indicated their inability to continue. The time needed to feel muscle fatigue was recorded using a digital stopwatch.
Given the possibility that this lower limb muscle fatigue protocol could exacerbate fatigue perception among the participants, the Borg Rating of Perceived Exertion was used to ensure that the participants did not indicate their perception of fatigue as their lower limb muscle fatigue. If the participants indicated their perception of fatigue before the lower limb muscle became fatigued, then they were allowed to rest. The lower limb muscle fatigue protocol was started again once their perception of fatigue was recovered. While the participants were performing the sit-to-stand task, their oxygen saturation level was monitored using a pulse oximeter. A small decrement in oxygen saturation level was allowed to stay at ≥94% throughout the fatigue protocol to ensure that the participants did not indicate their poor exercise tolerance as lower limb muscle fatigue. The assessor encouraged the participants to continue the sit-to-stand task if they were notified that their lower limbs had reached fatigue condition, but the oxygen saturation level was still above 94%. The walking task was carried out immediately after the fatigue protocol. The participants performed the extended TUG test immediately after the lower limb muscle fatigue protocol. The participants performed the walking task in less than 3 minutes after the fatigue protocol to prevent the full recovery of muscles.
Spatiotemporal gait parameters were captured using two digital video cameras. Two GoPro® Hero 4 Silver (GoPro Inc.) cameras were placed in the sagittal plane and used to capture data at 120 fps on a “normal” lens setting. Previous studies have suggested that digital video cameras, including GoPro cameras, can offer accurate and dependable measurements for gait analysis [25,26]. The video cameras were positioned on the right side of the participants at 5 m in the sagittal plane pathway. One video camera was positioned in the middle of the 7 m walking pathway, and another was set up at the turning area. The video cameras were mounted on a tripod to reduce the parallax error. The position of the video cameras was unaltered for the whole data collection process.
The recorded video was analyzed using a previously reported method [27]. Kinovea 0.8.15 (Kinovea.org) software was used to analyze the videos and determine step length and time. Stride length, stride time, and stride velocity were calculated using the following derived formulas: (1) stride length=left step length+right step length, (2) stride time=left step time+right step time, and (3) velocity=stride length/stride time.
The recorded videos were viewed and analyzed by two physiotherapists (mean years of working experience ≥9) with experience in gait evaluation of neurological patients to determine gait characteristics. A methodology published previously was used to describe the 180° turning characteristics [3]. The evaluators were given a form containing category definitions and descriptions for each level. The principal investigator discussed and clarified the keyword with the evaluators. The principal investigator also viewed the sample video with the evaluators to ensure it was analyzed according to category definitions and descriptions. The evaluators viewed the recorded video individually in random order to prevent bias. The analyses were organized according to (1) the time and number of steps taken to complete the extended TUG test and (2) the time and number of steps taken to accomplish the turning phase of the extended TUG test. Turning difficulty characteristics were evaluated according to (1) the presence or absence of staggering during the turn; (2) the type of turn or strategy used to accomplish the turn; (3) the number of steps or weight shifts taken during the turn; and (4) the amount of time taken to accomplish the turn. If the disagreement persists after discussion, a third reviewer or an expert in the field is brought in to independently evaluate the indicators of turning difficulty. The intra-rater reliability of the turning difficulty in our sample was determined to be good, with intraclass correlation coefficients ranging from 0.83 to 0.88.
Statistical analysis was conducted using IBM SPSS statistical version 26.0 (IBM Corp.). Descriptive statistics were calculated, and tests for normality were conducted for all outcome variables. Repeated-measures ANOVA was used to analyze gait parameters across condition (pre- and post-lower limbs fatigue protocol) and group (H&Y 2 and 3). Post-hoc Bonferroni comparison was performed when the repeated-measure ANOVA test revealed a significant difference (p<0.05). The turning difficulty characteristics were calculated in percentage.
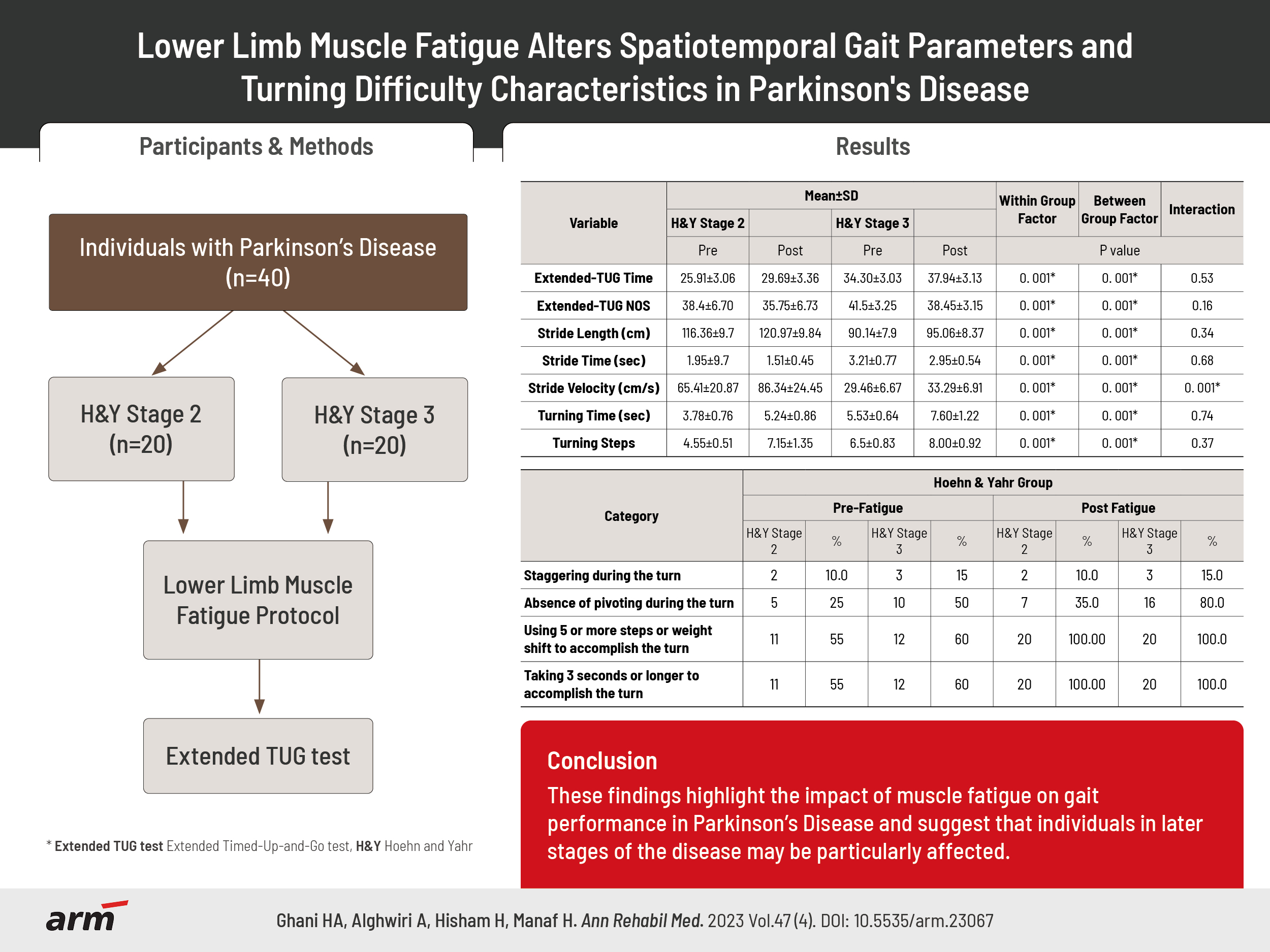
The characteristics of the participants are presented in Table 1. A total of 40 individuals with PD participated in this study, with 20 participants in H&Y stage 2 and 20 participants in H&Y stage 3. Out of the 40 participants, approximately 82.5% identified as male. No statistical differences were found in age, disease duration, height, weight, body mass index, and time to fatigue of the lower limb muscles between the two groups.
As shown in Table 2, individuals with PD in the H&Y stage 3 group required a longer time and a higher number of steps to complete the walking task (group effect, both p=0.001). The time and the number of steps to complete the extended TUG task significantly increased after lower limb muscle fatigue (condition effect, both p=0.001). The effect was similar for both groups (condition×group interaction, p>0.05).
Individuals with PD in the H&Y stage 3 group presented significantly shorter stride length, longer stride time, and slower stride velocity during the walking task after lower limb muscle fatigue condition (group effect, all p=0.001) compared with those in the H&Y stage 2 group. Significant increases were found in the stride length, decreased stride time, and reduced stride velocity during the walking task after lower limb muscle fatigue (condition effect, all p=0.001). The effect on stride length and stride time was similar for both groups (condition×group interaction, p>0.05). However, significant interaction effect was detected for stride velocity between both groups (condition×group interaction, p=0.001).
Individuals with PD in the H&Y stage 3 group required significantly longer time and more steps to accomplish the turn during the extended TUG test (group effect, both p=0.001). The time and number of steps to accomplish turning the during extended TUG test significantly increased after lower limb muscle fatigue (condition effect, both p=0.001). The effect was similar for both groups (condition×group interaction, p>0.05).
As shown in Table 3, only two to three participants for each group showed staggering during the turn. Individuals with PD in the H&Y stage 3 group were presented with an absence of pivoting during the turn (80.0%) compared with those in the H&Y stage 2 group (35.0%). Both groups took more steps and time to accomplish the turn, indicating turning difficulty (100%).
This study aimed to determine the effects of lower limb muscle fatigue on spatiotemporal gait parameters and turning difficulty indicators during the extended TUG test among individuals with PD in H&Y stages 2 and 3. We found several important findings. First, the PD groups showed significant spatiotemporal gait parameter changes under lower limb muscle fatigue conditions. Second, the stride velocity and length significantly increased after lower limb muscle fatigue. Third, the fatigue-related gait changes were more remarkable in individuals with PD in the H&Y stage 3 group. Lastly, both groups presented with turning difficulty; that is, they had nearly more than five steps to accomplish a 180° turn, and it took them more than 3 seconds to accomplish.
Individuals with PD in H&Y stages 2 and 3 experienced fatigue-related changes in spatiotemporal gait parameters, including increased stride length, decreased stride time, and reduced stride velocity during the walking task after lower limb muscle fatigue. This finding is possibly due to general hip and knee weakness and decreased ankle torque generation in the ankle among individuals with PD compared with controls [28]. These gait parameter changes could be an attempt to improve balance control in the anterior-posterior direction. The anterior margin of safety is negative during walking, which would cause a forward fall if the base of the support is not shifted forward by taking a further step [29]. Furthermore, increasing the step length during gait may retain the center of mass on the base of the support by decreasing the magnitude of the negative margin, making it easier to stop in a single step and avoid falling forward [29,30]. Similar changes in stride length after lower limb muscle fatigue were found in related studies [31,32]. Although the increase in stride length was small (~4 cm), it is still clinically relevant to individuals with PD. The possible explanation for this finding is that longer strides require more muscle activation, which could be a problem due to muscle weakness in this population [10].
Interestingly, the results reveal that the stride velocity and length significantly increased after lower limb muscle fatigue. The results are in line with the previous study that found both healthy individuals and patients with PD increased stride length and velocity after lower limb muscle fatigue [12]. Additionally, muscle fatigue decreased stride length in young adults and increased stride length in older adults [33]. This could be due to the fact that the body often employs compensation mechanisms to overcome muscle fatigue and maintain performance. These compensatory strategies may involve recruiting additional muscles or modifying movement patterns to generate the necessary force and velocity. Fatigue can also alter the biomechanics of movement, potentially leading to a stiffer and more propulsive gait pattern, allowing for a faster stride velocity. Additionally, muscle fatigue can decrease the ability of lower limb muscles to absorb and dissipate forces during the stance phase of gait, leading to a compensatory adoption of a stiffer and more propulsive gait pattern [34].
The interaction between the PD stage and fatigue can indeed have a significant impact on stride velocity. PD is a progressive neurodegenerative disorder that affects the motor system responsible for controlling movement. Fatigue is a common symptom experienced by individuals with PD, which can further exacerbate the motor difficulties associated with the condition [35]. Additionally, the slow and shuffling gait in PD is associated with an inability to generate an appropriate stride length, while the cadence remains normal or increased to reach a normal gait velocity. Fatigue in PD can further contribute to gait dysfunction, as patients with PD are more susceptible to muscle fatigue, which can damage their gait [12]. Therefore, the PD stage and fatigue combination can significantly impact stride velocity in individuals with PD.
In this study, individuals with PD in the H&Y stage 3 group were presented with shorter stride lengths than those in the H&Y stage 2 group. This finding is expected because quadriceps muscle strength and activation deficits correlate strongly with PD severity [10]. Although the fatigue protocol used is most likely to affect the entire leg muscles and not specific muscles, it could affect mainly the quadriceps muscles. As reported in a previous study using a similar fatigue protocol, the knee extension strength was reduced after the lower limb muscle fatigue protocol [36]. Another study that used a similar fatigue protocol reported a marked decrease in lower limb muscle strength among individuals with PD in a fatigued state [37]. Therefore, small adjustments are clinically essential to avoid falls during walking under the fatigued muscle condition.
The stride time decreased with lower limb muscle fatigue. Previous work reported similar trends, in which individuals with PD had reduced stride time while walking in a fatigued state [12]. The stride time decreased under external perturbations [38]. Furthermore, reducing the parameter would enhance the center of mass control and promote postural control in the anteroposterior and mediolateral directions, which appear to be the preferred strategy to deal with balance threats [38].
Increased stride length and reduced stride duration contributed to increased walking speed, possibly leading to more gait stability [39]. This finding broadly supports the work of another study; however, the mean stride velocity in the present study was considerably slower than that in a previous study (132.26±22.85) [12]. In line with the present findings, the gait speed with muscle fatigue increased in healthy older people [33]. Increased gait speed is associated with an increased risk of falls. However, the changes in stride length and duration are more critical in balance control than the resulting gait speed [38], and the stability of the gait pattern may be improved with a higher gait speed [39].
A significant increase in the number of steps and a longer time to complete the turning were observed among individuals with PD in this study. Similarly, previous work reported that the dynamic stability of individuals with PD was significantly smaller during fast turning. Thus, more time is taken for more dynamic stability during turning [40]. The same study reported that individuals with PD can turn more accurately at slow and preferred speeds. Another research indicated that individuals with early untreated PD can have a normal duration of the TUG test even though the 180° turn within the test was significantly slower than normal [4].
The timing deficit among the PD population is caused by faulty control of bilateral coordination [41]. Furthermore, a longer time to complete a turn is linked with freezing episodes [42]. However, none of the individuals with PD in the present study showed freezing episodes during walking and turning. Above all, the slower turning speeds of individuals with PD during turning angles might reflect a compensatory strategy to prevent dynamic postural instability [40]. Another study highlighted that the typical gait features of PD are more pronounced during turning than forward walking, especially in confined environments [43].
This study found that individuals with PD have been observed to experience increased turning difficulty following fatigue-induced fatigue simulation. Several studies have reported that fatigue can exacerbate motor symptoms in individuals with PD, including difficulties with turning [44,45]. They significantly took ≥5 steps to accomplish a turn under lower limb muscle fatigue conditions. Consistently, previous findings showed that individuals with PD without lower limb muscle fatigue took >4 steps to complete a turn [46]. The same study reported that individuals with PD have a longer turn time (>2 seconds) without being influenced by lower limb muscle fatigue. Moreover, individuals with PD in this study took ≥3 seconds to complete the turning task under lower limb muscle fatigue condition.
Although the results are relevant, this study has certain limitations that should be addressed. Several methodological factors should be considered because they can limit the capacity of the study to compare the findings with previous articles. First, the lack of control groups may hinder some statements about whether changes occur due to the fatigue protocol or another uncontrolled factor (i.e., learning effect or the time duration of experimental procedures). Practice learning effects may have occurred before or after testing. Second, the reduced time for individuals with PD to perform the lower limb muscle fatigue protocol may have limited the ability to draw meaningful conclusions. The reduced time could have led to an exacerbated fatigue perception and may not have been sufficient to cause muscle overload and induce metabolic fatigue. PD-related apathy or fear of post-exercise soreness or tiredness was a strategy that enabled them to achieve lower limb muscle fatigue more quickly during the fatiguing session without providing the researcher with a clear indication that they were acutely fatigued. Third, other lower limb muscles may be involved and have been fatigued. Even though the protocol most likely affected the entire legs and not just the specific muscles, the fatigue of other leg muscles, e.g., ankle muscles (triceps surae muscle), might have other (more pronounced) effects. Fourth, the study’s use of a GoPro camera instead of a force plate and three-dimensional camera may not adequately capture the complexities of gait patterns and mechanics, making applying the study's results to broader contexts challenging.
In conclusion, lower limb muscle fatigue affects spatiotemporal gait parameters among individuals with PD. Both groups demonstrated gait parameter changes that could be viewed as an attempt to improve balance and walking safety following lower limb muscle fatigue. Results indicated that individuals with PD in the H&Y stage 3 group had more difficulty turning while walking under lower limb muscle fatigued states than in the H&Y stage 2 group. These findings indicated an attempt to preserve balance and safety, probably to counteract the adverse effects of lower limb muscle fatigue. However, the gait adjustments could expose individuals with PD to a high risk of falling. Furthermore, the findings, coupled with a study reporting on the potential alteration of postural control in fatigued states among individuals with PD, strongly indicate that lower limb muscle fatigue has a measurable clinical effect on stability and potentially on the risk of falls among individuals with PD.
FUNDING INFORMATION
The authors thank the Universiti Teknologi MARA for funding the research project through the Geran Penyelidikan Khas [600-RMC/GPK 5/3 (134/2020)].
AUTHOR CONTRIBUTION
Conceptualization: Abd Ghani H. Methodology: Abd Ghani H, Manaf H. Formal analysis: Abd Ghani H. Funding acquisition: Manaf H. Project administration: Abd Ghani H. Writing – original draft: Abd Ghani H, Manaf H. Writing – review and editing: all authors. Approval of final manuscript: all authors.
Table 1.
Demographic data of the participants
Table 2.
Spatiotemporal gait parameters during extended TUG test
Table 3.
Indicators of turning difficulty
REFERENCES
1. Crenna P, Carpinella I, Rabuffetti M, Calabrese E, Mazzoleni P, Nemni R, et al. The association between impaired turning and normal straight walking in Parkinson's disease. Gait Posture 2007;26:172-8.


2. Patla AE, Adkin A, Ballard T. Online steering: coordination and control of body center of mass, head and body reorientation. Exp Brain Res 1999;129:629-34.



3. Thigpen MT, Light KE, Creel GL, Flynn SM. Turning difficulty characteristics of adults aged 65 years or older. Phys Ther 2000;80:1174-87.



4. Zampieri C, Salarian A, Carlson-Kuhta P, Aminian K, Nutt JG, Horak FB. The instrumented timed up and go test: potential outcome measure for disease modifying therapies in Parkinson's disease. J Neurol Neurosurg Psychiatry 2010;81:171-6.


5. Radder DLM, Sturkenboom IH, van Nimwegen M, Keus SH, Bloem BR, de Vries NM. Physical therapy and occupational therapy in Parkinson’s disease. Int J Neurosci 2017;127:930-43.



6. Lord S, Baker K, Nieuwboer A, Burn D, Rochester L. Gait variability in Parkinson’s disease: an indicator of non-dopaminergic contributors to gait dysfunction? J Neurol 2011;258:566-72.



7. Schlachetzki JCM, Barth J, Marxreiter F, Gossler J, Kohl Z, Reinfelder S, et al. Wearable sensors objectively measure gait parameters in Parkinson’s disease. PLoS One 2017;12:e0183989.



8. Orcioli-Silva D, Barbieri FA, Dos Santos PCR, Beretta VS, Simieli L, Vitorio R, et al. Double obstacles increase gait asymmetry during obstacle crossing in people with Parkinson's disease and healthy older adults: a pilot study. Sci Rep 2020;10:2272.




9. Murdock GH, Hubley-Kozey CL. Effect of a high intensity quadriceps fatigue protocol on knee joint mechanics and muscle activation during gait in young adults. Eur J Appl Physiol 2012;112:439-49.



10. Stevens-Lapsley J, Kluger BM, Schenkman M. Quadriceps muscle weakness, activation deficits, and fatigue with Parkinson disease. Neurorehabil Neural Repair 2012;26:533-41.



11. Rochester L, Jones D, Hetherington V, Nieuwboer A, Willems AM, Kwakkel G, et al. Gait and gait-related activities and fatigue in Parkinson’s disease: what is the relationship? Disabil Rehabil 2006;28:1365-71.


12. Santos PC, Gobbi LT, Orcioli-Silva D, Simieli L, van Dieën JH, Barbieri FA. Effects of leg muscle fatigue on gait in patients with Parkinson's disease and controls with high and low levels of daily physical activity. Gait Posture 2016;47:86-91.


13. Huang YZ, Chang FY, Liu WC, Chuang YF, Chuang LL, Chang YJ. Fatigue and muscle strength involving walking speed in Parkinson's disease: insights for developing rehabilitation strategy for PD. Neural Plast 2017;2017:1941980.




14. Baer M, Klemetson B, Scott D, Murtishaw AS, Navalta JW, Kinney JW, et al. Effects of fatigue on balance in individuals with Parkinson disease: influence of medication and brain-derived neurotrophic factor genotype. J Neurol Phys Ther 2018;42:61-71.



15. Weiss A, Mirelman A, Giladi N, Barnes LL, Bennett DA, Buchman AS, et al. Transition between the timed up and go turn to sit subtasks: is timing everything? J Am Med Dir Assoc 2016;17:864.e9-15.


16. Greene BR, Doheny EP, O'Halloran A, Anne Kenny R. Frailty status can be accurately assessed using inertial sensors and the TUG test. Age Ageing 2014;43:406-11.


17. Podsiadlo D, Richardson S. The timed “Up & Go”: a test of basic functional mobility for frail elderly persons. J Am Geriatr Soc 1991;39:142-8.


18. Rogers ME, Rogers NL, Takeshima N, Islam MM. Methods to assess and improve the physical parameters associated with fall risk in older adults. Prev Med 2003;36:255-64.


19. Shumway-Cook A, Brauer S, Woollacott M. Predicting the probability for falls in community-dwelling older adults using the Timed Up & Go Test. Phys Ther 2000;80:896-903.



20. Evans T, Jefferson A, Byrnes M, Walters S, Ghosh S, Mastaglia FL, et al. Extended “Timed Up and Go” assessment as a clinical indicator of cognitive state in Parkinson's disease. J Neurol Sci 2017;375:86-91.


21. Bedoya-Belmonte JJ, Rodríguez-González MDM, González-Sánchez M, Pitarch JMB, Galán-Mercant A, Cuesta-Vargas AI. İnter-rater and intra-rater reliability of the extended TUG test in elderly participants. BMC Geriatr 2020;20:56.




22. Wüest S, Massé F, Aminian K, Gonzenbach R, de Bruin ED. Reliability and validity of the inertial sensor-based Timed “Up and Go” test in individuals affected by stroke. J Rehabil Res Dev 2016;53:599-610.


23. Carvalho DV, Santos RMS, Magalhães HC, Souza MS, Christo PP, Almeida-Leite CM, et al. Can fatigue predict walking capacity of patients with Parkinson’s disease? Arq Neuropsiquiatr 2020;78:70-5.


24. Salarian A, Horak FB, Zampieri C, Carlson-Kuhta P, Nutt JG, Aminian K. iTUG, a sensitive and reliable measure of mobility. IEEE Trans Neural Syst Rehabil Eng 2010;18:303-10.



25. Hickey A, Del Din S, Rochester L, Godfrey A. Detecting free-living steps and walking bouts: validating an algorithm for macro gait analysis. Physiol Meas 2017;38:N1-15.



26. Sun J, Liu YC, Yan SH, Wang SS, Lester DK, Zeng JZ, et al. Clinical gait evaluation of patients with lumbar spine stenosis. Orthop Surg 2018;10:32-9.




27. Aung N, Bovonsunthonchai S, Hiengkaew V, Tretriluxana J, Rojasavastera R, Pheung-Phrarattanatrai A. Concurrent validity and intratester reliability of the video-based system for measuring gait poststroke. Physiother Res Int 2020;25:e1803.



28. Skinner JW, Lee HK, Roemmich RT, Amano S, Hass CJ. Execution of activities of daily living in persons with Parkinson disease. Med Sci Sports Exerc 2015;47:1906-12.


29. Hof AL, van Bockel RM, Schoppen T, Postema K. Control of lateral balance in walking. Experimental findings in normal subjects and above-knee amputees. Gait Posture 2007;25:250-8.


31. Barbieri FA. Impact of muscle fatigue on mechanics and motor control of walking [dissertation] Amsterdam, Vrije Universiteit Amsterdam. 2013.
32. Barbieri FA, dos Santos PC, Simieli L, Orcioli-Silva D, van Dieën JH, Gobbi LT. Interactions of age and leg muscle fatigue on unobstructed walking and obstacle crossing. Gait Posture 2014;39:985-90.


33. Granacher U, Wolf I, Wehrle A, Bridenbaugh S, Kressig RW. Effects of muscle fatigue on gait characteristics under single and dual-task conditions in young and older adults. J Neuroeng Rehabil 2010;7:56.




34. Huang S, Cai S, Li G, Chen Y, Ma K, Xie L. sEMG-based detection of compensation caused by fatigue during rehabilitation therapy: a pilot study. IEEE Access 2019;7:127055-65.

35. Alves G, Wentzel-Larsen T, Larsen JP. Is fatigue an independent and persistent symptom in patients with Parkinson disease? Neurology 2004;63:1908-11.


36. Barbieri FA, dos Santos PC, Vitório R, van Dieën JH, Gobbi LT. Effect of muscle fatigue and physical activity level in motor control of the gait of young adults. Gait Posture 2013;38:702-7.


37. Alota Ignacio Pereira V, Augusto Barbieri F, Moura Zagatto A, Cezar Rocha Dos Santos P, Simieli L, Augusto Barbieri R, et al. Muscle fatigue does not change the effects on lower limbs strength caused by aging and Parkinson's disease. Aging Dis 2018;9:988-98.



38. Hak L, Houdijk H, Steenbrink F, Mert A, van der Wurff P, Beek PJ, et al. Speeding up or slowing down?: gait adaptations to preserve gait stability in response to balance perturbations. Gait Posture 2012;36:260-4.


39. Bruijn SM, van Dieën JH, Meijer OG, Beek PJ. Is slow walking more stable? J Biomech 2009;42:1506-12.


40. Mellone S, Mancini M, King LA, Horak FB, Chiari L. The quality of turning in Parkinson’s disease: a compensatory strategy to prevent postural instability? J Neuroeng Rehabil 2016;13:39.




41. Plotnik M, Giladi N, Hausdorff JM. Bilateral coordination of gait and Parkinson’s disease: the effects of dual tasking. J Neurol Neurosurg Psychiatry 2009;80:347-50.


42. Hausdorff JM, Balash J, Giladi N. Effects of cognitive challenge on gait variability in patients with Parkinson’s disease. J Geriatr Psychiatry Neurol 2003;16:53-8.



43. Morris ME, Huxham F, McGinley J, Dodd K, Iansek R. The biomechanics and motor control of gait in Parkinson disease. Clin Biomech (Bristol, Avon) 2001;16:459-70.


44. Rahman S, Griffin HJ, Quinn NP, Jahanshahi M. Quality of life in Parkinson's disease: the relative importance of the symptoms. Mov Disord 2008;23:1428-34.


- TOOLS