- Search
| Ann Rehabil Med > Volume 46(6); 2022 > Article |
|
Abstract
Objective
To determine the appropriate time points to start regular exercise which could reduce age-related anxiety and impaired social behavior.
Methods
For this study, 8-week-old male Wistar rats were divided into three groups: no exercise (NoEX), short-term exercise (S-Ex), and long-term exercise (L-Ex) groups. S-Ex-group rats started treadmill exercise at 12 months of age, while L-Ex rats started from at 2 months of age. Exercise rats were forced to walk on the treadmill three times per week, with 1- to 2-day intervals for 10 minutes during the first 2 weeks, at 10 m/min until 17 months of age, and at 8 m/min thereafter. At 19 months of age, behavioral tests were performed to assess the effects of exercise on age-induced behavioral change as well as quantitative polymerase chain reaction were done to uncover the mechanism behind the behavioral changes.
Results
Anxiety-like behavior was improved by long-term exercise. Additionally, rats belonging to the S-Ex and L-Ex groups showed improved social behavior and increased curiosity about interesting objects. The qPCR data showed that treadmill exercise suppressed the expression of immediate-early genes in the prefrontal cortex of the aged rats.
Conclusion
This study suggests that long-term exercise represses early response genes, and in this way, it increases resistance to stress, diminishes anxiety-related behavior, and improves social behavior. These findings underscore the need to consider appropriate time to start exercise to prevent stress induced anxiety related behavior.
Aging results from the accumulation of molecular and cellular damage over time. In recent lifestyles, the reduced physical and mental function makes aging more susceptible to various illnesses and significantly impairs activities of daily living and quality of life. However, the average life expectancy of a person is over 60 years old, and the world population of this age group (over 60 years old) is expected to reach 2 billion by 2050 [1]. Therefore, developing effective strategies for delaying aging and averting aging-related illnesses is a pressing issue.
Physical exercise is widely accepted for preventing the deterioration of physical function and extending a healthy life [2,3]. Physical exercise improves both mental and physical health by exerting effects against depression [4], insomnia [5], and motor [6] and cognitive dysfunction [7]. Therefore, exercise is the best way to prevent various diseases caused by aging. So far, there are papers that have verified the effects of various exercises using animals. It has been reported that physical exercise plays a critical role in the survival of newly born hippocampal neurons in mice [8] and improves spatial cognitive function in rats [9]. Based on accumulating evidence, the effect of exercise against aging varies depending on the start time, schedule, and duration [10]. Focusing on a precise schedule, aerobic training on rats for 24 and 64 weeks showed a higher cognitive performance as evaluated by the Radial maze [11]. However, long-term forced treadmill exercise at 22 m/min for 1 hour (for 6 days per week) has been reported to improve cognitive function in young adults but not in aged rats [12]. In addition to this, there is a possibility that the effect of exercise may be compromised for inappropriate timing for commencing exercise.
Various basic studies have been conducted so far on the effects of aging and exercise. As described before, 18 years old in humans is equivalent to 6 months old in rats. Additionally, 45 and 60 years old in human are equivalent to 18 and 24 months old in rats, respectively [13]. Lifelong aerobic training showed that regular and moderate treadmill running has an anxiolytic effect on aged rats [11]. Another study reported that only 15 months of exercise in old rats had attenuative effects on age-induced hippocampal neuronal apoptosis and down-regulation of the Wnt signaling pathway [14]. Thus, the effects of exercise on aged rats are mostly related to hippocampal neuron for cognitive performance. Nevertheless, the effectiveness of exercise has also been recognized in the prefrontal cortex (PFC), which controls decision-making, attention, emotion, and social behavior. For example, a social interaction test to assess impulsivity was performed in attention-deficit hyperactivity disorder (ADHD) rats, and treadmill exercise attenuated hyper social behavior in ADHD rats [15]. Especially, social behavior declined with aging, and was suppressed in both male and female rats at 18 and 24 months [16]. However, there are no papers examining the effects on exercise in aged animals.
Therefore, here, we established two exercise paradigms to understand when it is appropriate to start exercising: a group that starts exercising from the beginning and a group that starts exercising midway through.
All animal experiments were performed following the Guidelines for Animal Experimentation of our institute and under the approval of the committee (Approval No. 05-U-31-2). Male Wistar rats were housed in pairs under-standard laboratory conditions.
Thirty-two 8-week-old male Wistar rats were housed by two animals per cage were randomly divided into three groups: non-exercise (NoEx; n=12), and short-term (S-Ex; n=10) and long-term (L-Ex; n=10) exercise groups. Treadmill exercise were implemented on S-Ex group rats from 12-month-old and L-Ex rats from 2-month-old. Rats were driven to walk on the belt using an electric shock (50 V alternating current) from a shock grid at the edge of the belt. Rats were forced to walk on a treadmill three times per week for 10 minutes for the first 2 weeks and thereafter for 20 minutes. The speed of the treadmill belt was 10 m/min until the rats became 17-month-old, and 8 m/min thereafter. NoEx group rats was kept in a cage without being placed on the treadmill. The numbers of animals were subjected to behavioral analyses were 11 rats (NoEx), 8 rats (L-Ex) and 8 rats (S-Ex group) as some rats were died for age-related health issues (Fig. 1).
Behavioral tests (light/dark box, social interaction, and sociosexual tests) were started from the 19 months, and were performed between 19:00 and 22:00.
To evaluate the effects of treadmill exercise on age-induced anxiety-like behavior, we employed the light and dark box test [17]. A plastic box with two chambers (40 cmГ—21 cmГ—21 cm, lengthГ—widthГ—height), one of which is covered with black tape (dark chamber), as illustrated in Fig. 2Aa. There is a square passage with 10 cm height and width between the two chambers, and rats can freely go through the passage. The movements were video-recorded and the duration staying in the light chamber and the frequency entering the light chamber were manually measured [11].
As previously described, we set up a test to assess social interaction and curiosity about funny objects [18,19]. Social interaction test was conducted using a rectangular field 20 cmГ—100 cm wide and 45 cm high which was surrounded by a wall and equipped with a video-tracking system. An aged rat was placed in the center, and the movement in the field was recorded for 5 minutes with a camera, and the number of times the rat entered the 20 cmГ—40 cm zone provided on the left and right was counted. Cylindrical boxes were set up in the left and right zones, and the zone containing small moving toys in the box was designated as the Toy zone, and the zone containing the unfamiliar 8-week-old male rat was designated as the Rat zone as described in Fig. 2Ba.
We examined social behavior with females as described elsewhere [20,21]. Here the test subject from each group was released into a 50 cm2 field with an unfamiliar 8-week-old female Wistar rat, where they were allowed to intercommunicate freely for 5 minutes. The duration of body contact and mounting made by aged rats were counted from the video recorder, as described in Fig. 2Ca.
RatsвҖҷ brains were dissected and homogenized, from which total RNA was extracted using a RNeasy Lipid Tissue Mini Kit (Qiagen, Hilden, Germany), as previously described [22]. All gene-specific mRNA measurements were normalized to glyceraldehyde 3-phosphate dehydrogenase mRNA levels. All polymerase chain reaction (PCR) primer sequences are listed in Table 1.
Aged ratsвҖҷ prefrontal cortices (PFC) were dissected at 19 months for immunoblotting. Rectangularly dissected contralateral and ipsilateral brain tissue samples were homogenized with Laemmli sample solution containing 3% sodium dodecyl sulfate for immunoblotting. The blots were incubated with the antibodies (Cell Signaling Technology, Danvers, MA, USA) listed in Table 2. Immunoreactive bands were analyzed by densitometry as described previously [22].
To evaluate the effects of treadmill exercise on age-induced anxiety-like behavior, in this study, the NoEx rats stayed for a comparatively short time in the bright box, while L-Ex rats stayed significantly longer in the bright box than NoEx rats (p<0.05). Concomitantly, L-Ex rats had a significantly reduced dark box residence time (p<0.05) compared with that of the NoEx rats (Fig. 2Ab). However, the S-Ex rats did not show any difference in spending time in the light or dark box compared with the NoEx group.
We set up a test by placing an unfamiliar adult male rat on one side and moving the toy to the other side to justify whether we investigated exercise induces any improvement in aged rats' social and motivational behaviors. Intriguingly, L-Ex rats showed an increased duration in both the stranger rat and toy zones compared to the rats of NoEx. Similarly, compared to the NoEx rats, the S-Ex rats also showed increased duration in both stranger rat and toy zones (Fig. 2Bb).
We evaluate the impact of exercise on the sociosexual activities of aged rats, there was no significant difference in the mounting duration between the groups. S-Ex rats had significantly longer contact times with young female rats than NoEx rats (p<0.05); however, L-Ex rats showed no significant differences compared with the NoEx and S-Ex rats (Fig. 2Cb).
We investigated whether exercise can alter the age-induced increased expression of c-Fos (proto-oncogene that is expressed within some neurons following depolarization), early growth response 1 (egr-1), c-Jun, and FosB (heterodimerizes with Jun family proteins to form activator protein 1 [AP-1] complexes that bind to AP-1 sites in responsive genes). First, we discovered that L-Ex significantly reduced the expression of c-Fos, FosB, and c-Jun mRNA compared with that in NoEx (p<0.05). Moreover, the egr-1 mRNA expression was found to be lower in both the S-Ex and L-EX groups compared with that in the NoEX group (p<0.01) (Fig. 3). Next, we examined the phosphorylation of c-Fos protein in PFC in 19-month-old rats, and the expression of the phosphorylated c-Fos protein was significantly reduced in the exercised S-Ex and L-Ex rats compared with that in the NoEx rats (p<0.05) (Fig. 4).
The present study focused on comparative beneficial effects of two paradigms of long-term exercise on ageing using healthy rats where only anxiety-related behavior was ameliorated by early started long-term exercise. However, both early started and late started long-term exercise showed better social behavior and increased curiosity about interesting objects. qPCR data also showed that treadmill exercise suppressed her IEGs expression in the PFC of aged rats.
Aging is characterized by a decline in bodily capabilities, sensitivities, and energies. As we grow older, various signals are activated, causing chronic inflammatory conditions that can lead to long-term stress responses [23]. The fact is that the aged brain is similar to the stressed brain, where regular exercise, dietary restriction, and cognitive enrichment are considered critical approaches for hormesis, which increases cellular stress response and resistance to neurodegenerative disorders [24]. Moreover, hypothalamic-pituitary-adrenal axis dysfunction is apparent in aging, wherein social isolation is one of the most potent sources of chronic stress [25]. We sought to address these issues by implementing two different exercise schedule packages, namely, implementation of treadmill exercise at an early adult age (8 weeks) and at middle age (48 weeks) in rats, for tackling age-related anxiety and lack of social behavior as prophylactic measures. The effects of treadmill exercise were first assessed on the anxiety response through tests based on impulsive behavior that evoked a state of light-dark contrast [11]. Anxiolytics have been found to increase locomotion and time spent in the light zone, whereas anxiogenics decrease them [17]. Aged rats subjected to treadmill exercise starting at an early adult age spent more time in a light box, indicating that treadmill exercise starting at early adulthood reduced age-induced anxiety-like behaviors. These findings are consistent with previous research, in which aged rats with prolonged treadmill walking spent more time in the light chamber of a light box than non-exercised rats [11]. Nonetheless, prolonged treadmill exercise starting at middle age had no effect on the anxiety behavior. The current result confirms that prolonged exercise starting from early adulthood is an effective prophylaxis against age-induced anxiety-related disorders.
Aging increases the risk of loneliness in animals and humans, and is a critical factor that impacts social and explorative behaviors [26,27]. Social behavior declines with age and is suppressed in old rats [16]. A growing number of studies on the neuronal substrates for social avoidance points toward distinct neural circuits encoding predatory fear or auditory fear conditioning [28]. In addition to this, another study on 5-week-old rats, in which treadmill exercise reduced the impairment of stress-induced social interaction [29], motivated us to study whether our exercise schedule can impact the social behavior of aged rats or not. On the social interaction test in our current study, both aged rats with treadmill exercise schedule packages showed increased contact time with an unfamiliar rat. However, they also had increased contact time with toys. Collectively, the data presented here suggest that prolonged treadmill running ameliorated age-induced social behavioral impairments and increased curiosity about funny objects. Furthermore, the improvement in social behavior seems to be related to the reduction of anxiety-like behavior on the treadmill. Anxiety plays an important role in social behavior, as anxious people are likely to refrain from social interactions to communicate with strangers [30]. In other words, the reduction of anxiety-like behavior on the treadmill may have led to the improvement of social behavior. In addition to social interaction, aging significantly impacts sociosexual interaction, and experience age-related declines in sexual behavior [31]. We discovered that treadmill exercise improves sociosexual interaction when started at middle age.
From a molecular mechanistic perspective, in this study, we hypothesized that treadmill exercise might ameliorate anxiety and improve social interaction of aged rats by surprising expression of IEGs, which are a general term for Fos family genes (c-Fos, FosB, Fra-l, Fra2) and Jun family genes (c-Jun, JunB, JunD), that are immediately generated by various external stress stimuli [32,33]. Additionally, c-Fos, c-Jun, and egr-1 are inducible transcription factors that increase in response to the activation of the intracellular signaling cade. The expression of IEGs is also associated with the activation of downstream transcription factors [34]. The induced stress increases c-Fos expression in the prefrontal cortex, locus coeruleus, and hypothalamus, as well as enhances anxiety behavior and affects emotions [35,36]. To investigate the molecular mechanisms that may underlie the effects of both long-term and short-term exercise on the relief of age-related stress, we quantified the mRNA expression of IEGs as they are reported to be expressed upon stress stimuli [37]. Herein, the IEGs mRNA and phosphorylation of the c-Fos protein in PFC decreased in aged rats when treadmill running was started at an early adult age. Chronic stress due to aging affects the hypothalamic-pituitary-adrenal axis [25] and aging weakens the negative feedback suppression of corticotropin releasing hormone (CRH) secretion from the hypothalamus [38]. In addition to this exposing aged rats to battery of behavioral tests may trigger this process. Therefore, Aging is more likely to be stressed, which may elevate plasma corticosterone (CORT) levels. But wheel running may enhance negative feedback by altering the ratio of mineralocorticoid and glucocorticoid receptors (GR) in the hippocampus [39]. And, wheel-running mice reached peak CORT levels significantly earlier than sitting mice and subsequently more quickly reached baseline CORT levels. Exercise may produce a rapid adaptive response to stress by promoting negative feedback on the hypothalamic-pituitary-adrenal axis [40]. In particular, post-stress exercise suppresses elevated c-Fos, showing anxiolytic effects [36]. Therefore, long-term exercise may acquire stress tolerance and can be recommended as a preventive and therapeutic method for the effects of stress and anxiety. Collectively, these results suggest that treadmill exercise ameliorated age-induced anxiety and social behavioral impairment by inhibiting IEGs expression.
There are some limitations of this study. Considering the extent of the study, it is tough to perform further investigation to elucidate exact cellular mechanism behind the beneficial effects of long-term exercise. Furthermore, some factors such as tumors and age-related death plays depressing role behind this study.
In conclusion, this study suggests that long-term exercise acquires stress resistance by suppressing IEG in the PFC and improves anxiety-related and social behaviors. These results emphasize the need to consider the appropriate timing to prevent anxiety-related behaviors in stress.
AUTHOR CONTRIBUTION
Conceptualization: Tanaka J. Methodology: Utsunomiya R, Mikami K, Taguchi S. Formal analysis: Taguchi S, Choudhury ME. Funding acquisition: Choudhury ME. Project administration: Choudhury ME. Visualization: Utsunomiya R, Mikami K, Taguchi S. Intellectual Input: Yano H. Writing вҖ“ original draft: Taguchi S. Writing вҖ“ review and editing: Choudhury ME. Approval of final manuscript: all authors.
ACKNOWLEDGMENTS
This work was funded by Grants-in-Aid for Scientific Research of the Japan Society for the Promotion of Science (No. 17K16650 and 20K06856) awarded to Mohammed E. Choudhury.
Fig.В 1.
Experimental design. Thirty-two 2-month-old rats were randomly assigned to three groups, each containing 11Вұ1 rats: NoEx, non-exercized rats (n=12); S-Ex rat, exercise started at 12 months of age (n=10); and L-Ex rats, exercise started at 2 months of age (n=10). Rats belonging to the S-Ex and L-Ex groups exercised three times a week until 19 months of age. In this experiment, exercise was performed every other day or two to prevent the rats from being stressed or tired. Behavioral tests were performed after the exercise session, followed by brain sampling for qPCR (quantitative polymerase chain reaction) and immunoblotting. Considering the nocturnal nature of the animal, all experimental approaches were conducted from 19:00 to 22:00 to minimize the behavioral effects of diurnal cycles. LD box text, light/dark box text.
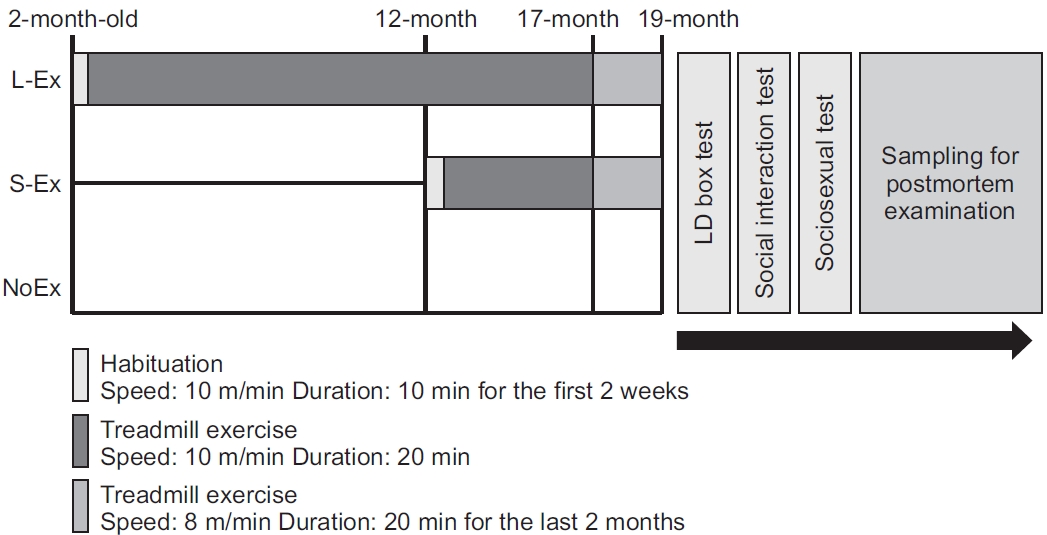
Fig.В 2.
Long-term forced treadmill exercise reduces anxiety-like and improves social behavior. (Aa) Layout for light/dark box test instrumental setting and procedure. (Ab) Bar graph for light and dark boxes, wherein rats from the L-Ex group spent more time in the light box and less time in the dark box than the NoEx group. (Ba) Layout for the instrumental properties for the social interaction test. (Bb) Bar graph for the social interaction test, wherein rats from both the S-Ex and L-Ex groups entered the stranger rat zone more frequently. (Ca) Layout for the instrumental stuff and experimental method. (Cb) Bar graph for the sociosexual interaction test, wherein rats belonging to the S-Ex group showed increased body sniffing duration, but there were no changes in mounting duration. Data (n=8вҖ“11) were presented as meanВұstandard deviation and analyzed with ANOVA and Tukey post-hoc test. *p<0.05, **p<0.01 indicate significant differences, and (ns) p>0.05 indicates nonsignificant differences between the two groups.
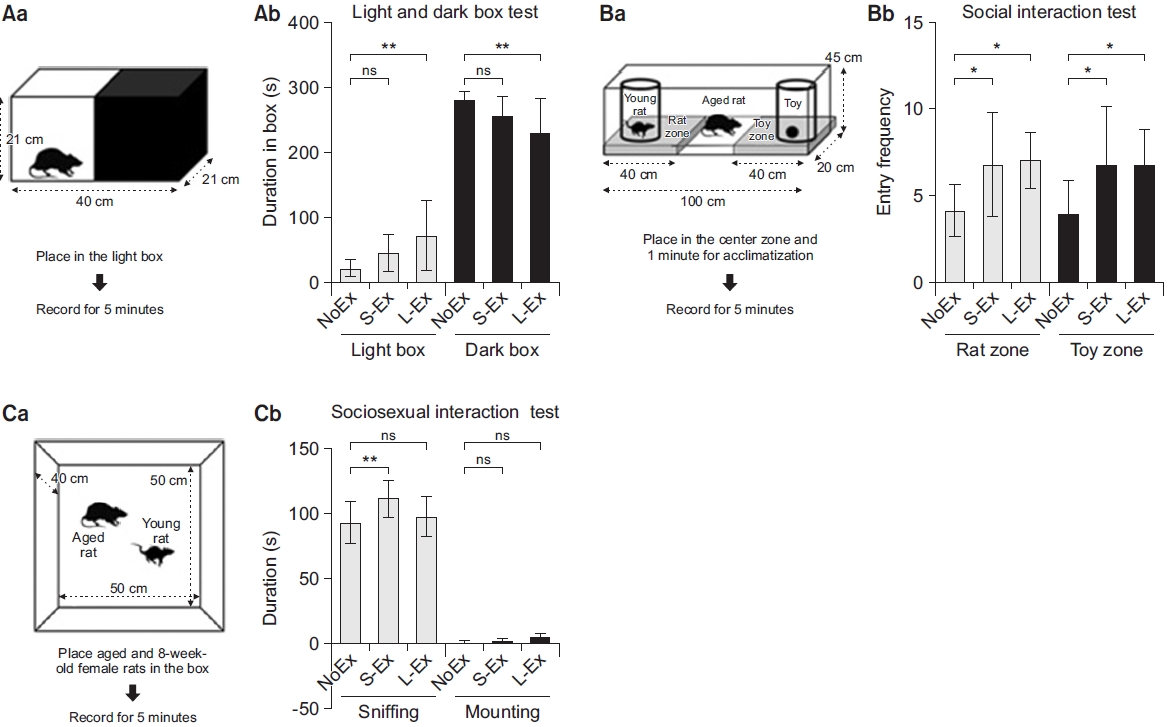
Fig.В 3.
Treadmill exercise inhibited IEG mRNAs expression on the prefrontal cortex tissue samples dissected at 19 months. Data shows the expression of mRNA encoding c-Fos, FosB, c-Jun, and egr-1. The black bar represents nonexercized (NoEx) rats, the light-gray bar represents short-term exercise (S-Ex) rats, and the dark gray bar represents long-term exercise (L-Ex) rats. Data (n=5; NoEx, S-Ex, and L-Ex) are shown as meanВұstandard deviation. Data (n=5) were analyzed with ANOVA and Tukey post-hoc test. *p<0.05, **p<0.01 indicate significant differences, and (ns) p>0.05 indicates non-significant differences between the two groups. IEG, immediate early gene.
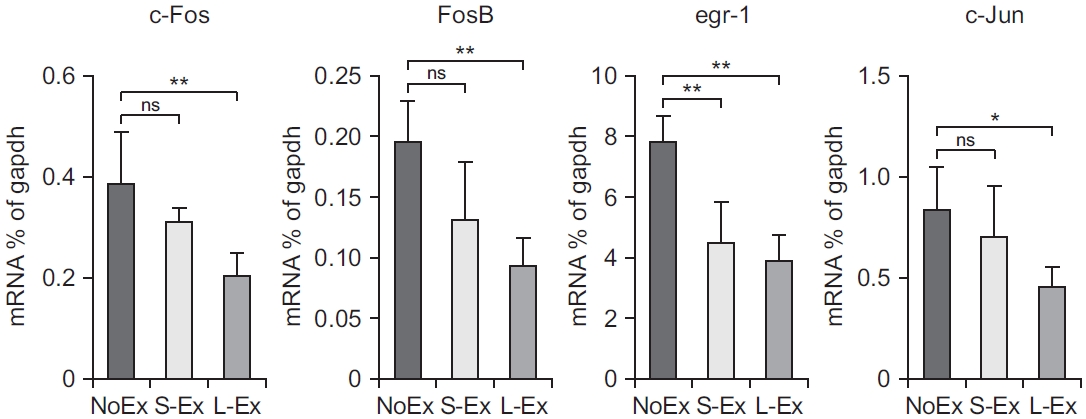
Fig.В 4.
Long-term exercise increased the phosphorylation of c-Fos at the prefrontal cortex (PFC) tissue. (Left) Representative immunoblotting image, showing the total c-Fos; and phosphorylated c-Fos, showing the expression changes in the protein levels at the PFC of 19-monthold NoEx, S-Ex, and L-Ex rats, respectively. (Right) Ratio of densitometric analysis of the total and phosphorylated c-Fos, shown as meanВұstandard deviation (n=5; NoEx, S-Ex, L-Ex). Black bar represents NoEx rats, lightgray bar represents S-Ex rats, and dark bar represents L-Ex rats. *p<0.05 vs. NoEx. Data (n=5) were analyzed with ANOVA and Tukey post-hoc test and are shown as meanВұstandard deviation.
REFERENCES
1. Beard JR, Officer A, de Carvalho IA, Sadana R, Pot AM, Michel JP, et al. The world report on ageing and health: a policy framework for healthy ageing. Lancet 2016;387:2145-54.


2. Cotman CW, Berchtold NC. Exercise: a behavioral intervention to enhance brain health and plasticity. Trends Neurosci 2002;25:295-301.


3. Billinger SA, Arena R, Bernhardt J, Eng JJ, Franklin BA, Johnson CM, et al. Physical activity and exercise recommendations for stroke survivors: a statement for healthcare professionals from the American Heart Association/American Stroke Association. Stroke 2014;45:2532-53.


4. Kandola A, Ashdown-Franks G, Hendrikse J, Sabiston CM, Stubbs B. Physical activity and depression: towards understanding the antidepressant mechanisms of physical activity. Neurosci Biobehav Rev 2019;107:525-39.


5. Reid KJ, Baron KG, Lu B, Naylor E, Wolfe L, Zee PC. Aerobic exercise improves self-reported sleep and quality of life in older adults with insomnia. Sleep Med 2010;11:934-40.



6. Rezaee Z, Marandi SM, Esfarjani F. Age-related biochemical dysfunction in 6-OHDA model rats subject to induced-endurance exercise. Arch Gerontol Geriatr 2022;98:104554.


7. O'Callaghan RM, Griffin EW, Kelly AM. Long-term treadmill exposure protects against age-related neurodegenerative change in the rat hippocampus. Hippocampus 2009;19:1019-29.


8. Fabel K, Wolf SA, Ehninger D, Babu H, Leal-Galicia P, Kempermann G. Additive effects of physical exercise and environmental enrichment on adult hippocampal neurogenesis in mice. Front Neurosci 2009;3:50.



9. Van der Borght K, Havekes R, Bos T, Eggen BJ, Van der Zee EA. Exercise improves memory acquisition and retrieval in the Y-maze task: relationship with hippocampal neurogenesis. Behav Neurosci 2007;121:324-34.


10. Vecchio LM, Meng Y, Xhima K, Lipsman N, Hamani C, Aubert I. The neuroprotective effects of exercise: maintaining a healthy brain throughout aging. Brain Plast 2018;4:17-52.



11. Pietrelli A, Di Nardo M, Masucci A, Brusco A, Basso N, Matkovic L. Lifelong aerobic exercise reduces the stress response in rats. Neuroscience 2018;376:94-107.


12. Asl NA, Sheikhzade F, Torchi M, Roshangar L, Khamnei S. Long-term regular exercise promotes memory and learning in young but not in older rats. Pathophysiology 2008;15:9-12.


14. Chen D, Zhang Y, Zhang M, Chang J, Zeng Z, Kou X, et al. Exercise attenuates brain aging by rescuing down-regulated Wnt/ОІ-catenin signaling in aged rats. Front Aging Neurosci 2020;12:105.



15. Baek DJ, Lee CB, Baek SS. Effect of treadmill exercise on social interaction and tyrosine hydroxylase expression in the attention-deficit/hyperactivity disorder rats. J Exerc Rehabil 2014;10:252-7.




16. Perkins AE, Doremus-Fitzwater TL, Spencer RL, Varlinskaya EI, Conti MM, Bishop C, et al. A working model for the assessment of disruptions in social behavior among aged rats: the role of sex differences, social recognition, and sensorimotor processes. Exp Gerontol 2016;76:46-57.



17. Bourin M, Petit-Demouliere B, Dhonnchadha BN, Hascoet M. Animal models of anxiety in mice. Fundam Clin Pharmacol 2007;21:567-74.


18. Kaidanovich-Beilin O, Lipina T, Vukobradovic I, Roder J, Woodgett JR. Assessment of social interaction behaviors. J Vis Exp 2011;(48):2473.

19. Clapcote SJ, Lipina TV, Millar JK, Mackie S, Christie S, Ogawa F, et al. Behavioral phenotypes of Disc1 missense mutations in mice. Neuron 2007;54:387-402.


20. Portillo W, Unda N, Camacho FJ, Sanchez M, Corona R, Arzate DM, et al. Sexual activity increases the number of newborn cells in the accessory olfactory bulb of male rats. Front Neuroanat 2012;6:25.



21. Hong W, Kennedy A, Burgos-Artizzu XP, Zelikowsky M, Navonne SG, Perona P, et al. Automated measurement of mouse social behaviors using depth sensing, video tracking, and machine learning. Proc Natl Acad Sci U S A 2015;112:E5351-60.



22. Taguchi S, Choudhury ME, Miyanishi K, Nakanishi Y, Kameda K, Abe N, et al. Aggravating effects of treadmill exercises during the early-onset period in a rat traumatic brain injury model: when should rehabilitation exercises be initiated? IBRO Rep 2019;7:82-9.



23. Chung HY, Kim DH, Lee EK, Chung KW, Chung S, Lee B, et al. Redefining chronic inflammation in aging and age-related diseases: proposal of the senoinflammation concept. Aging Dis 2019;10:367-82.



24. Mattson MP. Hormesis and disease resistance: activation of cellular stress response pathways. Hum Exp Toxicol 2008;27:155-62.



25. Steptoe A, Shankar A, Demakakos P, Wardle J. Social isolation, loneliness, and all-cause mortality in older men and women. Proc Natl Acad Sci U S A 2013;110:5797-801.



26. Friedman A, Hueske E, Drammis SM, Toro Arana SE, Nelson ED, Carter CW, et al. Striosomes mediate value-based learning vulnerable in age and a HuntingtonвҖҷs disease model. Cell 2020;183:918-34.e49.



27. Altun M, Bergman E, Edstrom E, Johnson H, Ulfhake B. Behavioral impairments of the aging rat. Physiol Behav 2007;92:911-23.


28. Silva BA, Mattucci C, Krzywkowski P, Murana E, Illarionova A, Grinevich V, et al. Independent hypothalamic circuits for social and predator fear. Nat Neurosci 2013;16:1731-3.




29. Kim TW, Lim BV, Kim K, Seo JH, Kim CJ. Treadmill exercise alleviates stress-induced impairment of social interaction through 5-hydroxytryptamine 1A receptor activation in rats. J Exerc Rehabil 2015;11:192-7.




30. Wu T, Luo Y, Broster LS, Gu R, Luo YJ. The impact of anxiety on social decision-making: behavioral and electrodermal findings. Soc Neurosci 2013;8:11-21.


31. Spruijt BM, Meyerson BJ, Hoglund U. Aging and sociosexual behavior in the male rat. Behav Brain Res 1989;32:51-61.


32. Senba E, Ueyama T. Stress-induced expression of immediate early genes in the brain and peripheral organs of the rat. Neurosci Res 1997;29:183-207.


33. Weinberg MS, Girotti M, Spencer RL. Restraint-induced fra-2 and c-fos expression in the rat forebrain: relationship to stress duration. Neuroscience 2007;150:478-86.


34. Perkins AE, Woodruff ER, Chun LE, Spencer RL, Varlinskaya E, Deak T. Analysis of c-Fos induction in response to social interaction in male and female Fisher 344 rats. Brain Res 2017;1672:113-21.



35. Bland ST, Schmid MJ, Der-Avakian A, Watkins LR, Spencer RL, Maier SF. Expression of c-fos and BDNF mRNA in subregions of the prefrontal cortex of male and female rats after acute uncontrollable stress. Brain Res 2005;1051:90-9.


36. Seo JH, Kim TW, Kim CJ, Sung YH, Lee SJ. Treadmill exercise during pregnancy ameliorates post-traumatic stress disorder-induced anxiety-like responses in maternal rats. Mol Med Rep 2013;7:389-95.


37. Kubik S, Miyashita T, Guzowski JF. Using immediate-early genes to map hippocampal subregional functions. Learn Mem 2007;14:758-70.


38. Smith MA. Hippocampal vulnerability to stress and aging: possible role of neurotrophic factors. Behav Brain Res 1996;78:25-36.


- TOOLS








