- Search
| Ann Rehabil Med > Volume 43(1); 2019 > Article |
|
Abstract
Objective
To elucidate the effect of task-specific lower extremity training (TSLET) on cognitive and gait function in stroke patients.
Methods
Thirty-eight patients were assigned to either the TSLET group or the control group. The individuals of TSLET group went through a TSLET plus conventional physical therapy. The control group received two sessions of conventional physical therapy. The primary outcome involved the assessment with visual and auditory digit span test. The secondary outcome was evaluated by the Korean version of Mini-Mental State Examination (K-MMSE) and Global Deterioration Scale (GDS) for cognitive function, Berg Balance Scale (BBS), Time Up and Go Test (TUG), 10 meters Walking Test (10mWT), 6 minutes Walking Test (6MWT), and Korean version of Modified Barthel Index (K-MBI) for gait, balance, and functional ability.
Results
After intervention (3 weeks) and 2 weeks of follow-up, the TSLET group showed statistically significant improvement in the visual digit span test backwards compared with the control group. In secondary outcome, a significant improvement was observed in GDS, BBS, TUG, and 10mWT in the TSLET group. There was no significant difference between the two groups concerning visual digit span test forward, auditory forward and backward digit span tests, K-MMSE, 6MWT, and K-MBI.
Cerebrovascular disease is an important cause of morbidity in the elderly, and it causes serious cognitive impairment as well as physical disability [1]. Up to 64% of patients who have experienced stroke have some degree of cognitive impairment [2]. Stroke-related cognitive deficits interfere with functional recovery and the potential benefits of rehabilitation [3]. Furthermore, the presence of cognitive impairment increases the risk of recurrent stroke, mortality, and diminishes the activities of daily living [3,4]. Interference in cognitive tasks and motor activity is important for functional improvement in patients with neurological deficits [5]. Therefore, it is necessary to consider both effective cognitive and motor training in rehabilitation therapy.
The positive effects of various training programs on cognition have been identified. Physical activities such as walking and jogging resulted in a reduced risk of cognitive impairment in stroke survivors [6]. In addition, resistance training led to enhancement in cognitive performance and maximized cognitive recovery after a stroke [7]. Tai Chi training, a kind of task-specific training that combines aspects of aerobic, strength, flexibility or balance training, had positive effects in both cognitively impaired adults and healthy older adults. This complex exercise program requiring diverse movements following instructions has demonstrated improvements in various fields such as global cognitive and executive functions, processing speed, attention, and working memories [8]. However, the Tai Chi training is not suitable for stroke patients, as it requires good functional level for independent movement. Another problem with stroke patients is that it is difficult to establish an exercise protocol for them due to multiple factors like aerobic, mobility moment, abdominal breathing, and relaxation exercises.
Task-specific lower extremity training (TSLET) is a complex lower extremity exercise during which subjects learn about lower extremity movement while solving a problem using visual feedback. The TSLET is a relatively safe and easy training method, which can be applied for stroke patients with low functional level. The previous study showed that the TSLET was effective in improving gait and balance compared to conventional physical therapy [9]. However, its effect on cognitive function has not been well established.
Since physical activities such as walking and taskoriented Tai chi training are known to improve cognitive function, we predicted that the TSLET would be likely effective in improving cognitive function in stroke patients.
While the previous study confirmed the effect of TSLET in gait and balance function for 2 weeks [9], the present study was carried out to clarify the training effect for a longer period of 3 weeks and for further 2 weeks for postponed effect.
We aimed to elucidate the effects of TSLET on cognitive and gait function in stroke patients in comparison with conventional physical therapy.
A randomized controlled and prospective study was carried out to investigate the effect of TSLET on cognitive and gait function in subacute stroke patients. Thirty-eight patients were randomly assigned to one of the two groups, the TSLET group (n=19) or the control group (n=19). Patients and the assessing physicians were blinded to group allocation [6,10].
Patients who had experienced stroke and had been admitted to the Department of Rehabilitation Medicine, Wonju Severance Christian Hospital between June and September 2017 were selected for the present study. These patients were diagnosed with stroke through computed tomography or magnetic resonance imaging. The patients were included in the study based on the following inclusion criteria: (1) age over 20 years, (2) subacute stroke patients within 6 months after stroke, (3) Berg Balance Scale (BBS) of 21-40, (4) patients who did not have a previous history of stroke, and (5) absence of severe musculoskeletal and cardiovascular disease. Patients with secondary brain injury such as recurrence of cerebral infarction or intracranial hemorrhage, hydrocephalus, aphasia, hemineglect, or visual field defect were excluded.
This study was approved by the Institutional Review Boards of Wonju Severance Christian Hospital (No. CR216001) and conducted according to the Declaration of Helsinki and in keeping with local regulations. Written informed consents were obtained from all participants in this study.
The TSLET group received one session of the TSLET (30 minutes) and one session of conventional physical therapy (30 minutes), 5 days a week for 3 weeks. The control group received two sessions of conventional physical therapy (one session, 30 minutes; total, 60 minutes), 5 days a week for 3 weeks. Subsequently, both the groups were equally trained with conventional physical therapy (30 minutes), 5 days a week for 2 weeks to evaluate postponed effect of TSLET. In both the groups, cognitive and gait functions were evaluated before and after 3 weeks and after 5 weeks of training.
We used 3DBT-12 machine (Man&Tel, Gumi, Korea) for TSLET. This device has two electronic body weight scales that can display the weight of both sides. It consists of a camera, a tilting sensor that can recognize the angle of the knee joint, screen to help with visual feedback, and computer system to enable game using measured information. When the body weight scale recognizes the degree of weight applied to the left and right sides, the values and graphs appear on the screen in real time, with kg unit marked until the second decimal place.
Pressure sensors recognize the pressure center and translate it into a horizontal cursor movement. A tilting sensor attached to the lateral side of knee measures knee joint angle and movement and translates the measurement into a vertical cursor movement on the screen. We used вҖҳfruit picking gameвҖҷ. The catching cursor moves up and down according to the flexion and extension of the knee joint and moves the cursor left and right through the weight shifting. The number of apples caught for a given period of time was converted into a percentage value to be measured as a score. To prevent falling down, harness and safety bar was used (Fig. 1). The difficulty of this intervention was adjusted according to the function of each patient.
The conventional physical therapy was based on a balance exercise program. The balance exercise focused on trunk stabilization, weight transfer, and strengthening and exercise with balance board for balance and proprioception. The training was guided by the physical therapists.
Outcome measurements for the primary outcome were the digit span test using computerized neuropsychological test (CoTras-P; MEDI-IS, Seoul, Korea) [11]. Digit span test mainly reflects the executive function among various cognitive functions. Visual digit span test visually presents the digit sequences, which are displayed simultaneously in a row. After looking at the number for 15 seconds, the participants were advised to wait for 3 seconds and then asked to call out the numbers in order either from left to right (forward) or from right to left (backward). This test was terminated when the participant failed to reproduce the lists [12]. In auditory span test, lists of digits were presented using live voice. After listening to the lists of numbers, the participants were asked to wait for 3 seconds and then call out the numbers in the list in either forward or backward order. Typically, the test starts with 3 numbers, increased by 5 numbers, and finished when the participant fails to reproduce the lists [13].
Secondary outcome measures were used to evaluate additional cognitive and physical function. The Korean version of Mini-Mental State Examination (K-MMSE) and Global Deterioration Scale (GDS) were used to evaluate cognitive function. K-MMSE has 7 domains, scored between 0 and 30, with a low score indicating poor cognitive function [14]. The K-MMSE assesses orientation for time and place, memory registration, and recall, attention/concentration, language (naming, repetition, threestage verbal command, writing skills, and visuospatial construction [15]. GDS is an instrument used to assess cognitive decline at seven different stages. Stage 1 indicates no cognitive decline, stage 2 reflects a very mild cognitive decline, and stages 3вҖ“7 define mild, moderate, moderately severe, severe, and very severe cognitive decline, respectively. From stage 5 and higher, an individual is unable to perform daily living independently [16].
BBS, Time Up and Go Test (TUG), 10 meters Walking Test (10mWT), 6 minutes Walking Test (6MWT), and Korean version of Modified Barthel Index (K-MBI) were measured to evaluate physical performance, including gait, balance, and functional status. BBS is an objective measurement of dynamic balance through 14 tasks and can detect minimal changes in stroke patients [17]. TUG is a single-item test that requires the subject to stand up, walk for 3 meters, turn back, and sit down again, and it is a reliable tool to evaluate functional ambulation status. The 10mWT is an assessment tool for functional mobility, especially maximal walking speed, by measuring the time taken to walk a 10-meter distance [18]. The 6MWT is a submaximal exercise test that evaluates functional endurance for a distance walked over a span of 6 minutes [19]. K-MBI measures the performance of the daily living activity and it is a reliable and valid assessment for measuring functional status in stroke patients [20].
All data were analyzed using SPSS version 19.0 (IBM Inc., New York, NY, USA). To compare the characteristics between the two groups, two-sample t-test or Wilcoxon rank sum test were used for continuous variables, and chi-square test or Fisher exact test was applied for categorical variables. For statistical analyses, a linear mixed model for a repeated-measures covariance pattern model with unstructured covariance within subjects was used. Two fixed effects were included: one between-subjects group effect (TSLET and control) and one within-subject time effect (three times: baseline, 3 weeks, and 5 weeks). Possible difference variables (visual digit span test forward [VST-F], visual digit span test backward [VST-B], auditory digit span test forward [AST-F], auditory digit span test backward [AST-B], K-MMSE, GDS, BBS, TUG, 10mWT, 6MWT, K-MBI) in the groups across time were analyzed according to timeГ—group interactions. When a significant difference between the two groups was noted across time, a post-doc Bonferroni multiple comparison test was used. A p-value less than 0.05 was considered significant.
Thirty-eight patients met the inclusion criteria and underwent randomization. However, 3 of 38 patients dropped out, 1 from the TSLET group and 2 from the control group due to follow-up loss. The remaining 35 participants in the TSLET and the control groups completed the entire study (Fig. 2).
The demographic and clinical characteristics of the patients are summarized in Table 1. There were no significant differences between the two groups in their gender, age, height, weight, duration of the stroke, paretic side, type of stroke, and National Institutes of Health Stroke Scale (NIHSS) score.
In the TSLET group, the baseline VST-B score in the TSLET group increased from 3.77Вұ1.60 to 4.49Вұ1.24 after intervention (3 weeks) and from 3.77Вұ1.60 4.59Вұ1.39 at 2 weeks of follow-up, respectively. VST-B score increased about 21.75% relative to baseline. In the control group, the VST-B score was 3.91Вұ1.45, which increased to 4.42Вұ1.19 after intervention and 4.21Вұ1.53 at 2 weeks of follow-up period; overall a 7.67% VST-B score change relative to baseline was noted. A significant time versus group interaction was observed for VST-B improvement (p=0.031) (Fig. 3). Post-hoc comparisons showed that the TSLET group exhibited greater improvement in VST-B than the control group after intervention (p=0.038) and at 2 weeks (p=0.032) of the follow-up period. However, no significant difference between the values after intervention and 2 weeks of the follow-up period (p=0.731) was observed. The baseline VST-F, AST-F, and AST-B scores in the TSLET group were 4.50Вұ1.29, 4.75Вұ1.10, and 3.01Вұ0.87, respectively and these scores increased to 4.86Вұ1.33, 5.23Вұ1.07, and 3.31Вұ0.78 after intervention and to 4.94Вұ1.24, 5.06Вұ1.21, and 3.30Вұ0.75 at 2 weeks of the follow-up period. In the control group, the baseline VST-F, AST-F, and AST-B scores were 4.66Вұ1.17, 5.36Вұ1.74, and 3.21Вұ0.99 and these scores changed to 4.76Вұ1.11, 4.9+Вұ1.36, and 3.38Вұ0.96 after intervention and to 4.82Вұ1.24, 4.96Вұ1.33, and 3.52Вұ1.05 at 2 weeks of the follow-up period. Results from the mixed model showed no significant time versus group interaction for changes in VST-F, AST-F, and AST-B scores (p=0.745, p=0.826, and p=0.671, respectively). Post-hoc comparisons also showed no significant improvement in the TSLET group compared to the control group after intervention (VST-F p>0.999, AST-F p=0.419, AST-B p>0.999) and at 2 weeks of follow-up period (VST-F p>0.999, AST-F p=0.892, AST-B p>0.999). No significant differences in the change in these scores were noted between the values after intervention and 2 weeks of the follow-up period (VST-F p>0.999, AST-F p=0.949, AST-B p>0.999).
The baseline GDS score in the TSLET group decreased from 3.44Вұ1.04 to 2.61Вұ1.09 after intervention and to 2.55Вұ1.12 at 2 weeks of the follow-up period, respectively (Table 2). In the control group, the baseline GDS score in the TSLET group decreased from 3.24Вұ1.03 to 2.94Вұ0.90 after intervention and to 2.77Вұ0.83 at 2 weeks of the follow-up period. Results from the mixed model showed a significant time versus group interaction for change in GDS (p=0.041). Post-hoc comparisons showed a significantly greater improvement in GDS in the TSLET group compared with the control group after intervention (p=0.037) and at 2 weeks (p=0.043) of follow-up. However, there was no significant improvement between 3 and 5 weeks of training effect (p=0.781).
After 5 weeks of training, the TSLET group exhibited significantly greater improvement in BBS, TUG, and 10mWT after training compared with the control group. The baseline BBS, TUG, and 10mWT in the TSLET group were 31.22Вұ9.36, 33.89Вұ18.00, and 23.86Вұ12.83, respectively and these scores changed to 39.19Вұ9.12, 25.13Вұ17.20, and 21.11Вұ12.06 after intervention and to 43.15Вұ7.98, 22.73Вұ16.30, and 18.65Вұ14.50 at 2 weeks of follow-up. In the control group, the baseline BBS, TUG, and 10mWT were 31.94Вұ9.36, 27.93Вұ18.00, and 19.33Вұ12.83 and these scores changed to 34.94Вұ8.84, 25.47Вұ16.70, and 21.11Вұ12.06 after intervention and to 36.59Вұ9.57, 26.88Вұ18.75, and 21.34Вұ13.22 at 2 weeks of follow-up. Results from the mixed model showed a significant time versus group interaction for changes in BBS (p=0.004), TUG (p<0.001), and 10mWT (p<0.001). Post-hoc comparisons revealed that the TSLET group exhibited a significantly greater improvement in BBS, TUG, and 10mWT after intervention (BBS p=0.017, TUG p=0.042, 10mWT p=0.032) and at 2 weeks of follow up (BBS p=0.003, TUG p=0.004, 10mWT p<0.001) compared with the control group. The TUG and 10mWT showed a significant difference between the values after intervention and at 2 weeks of follow-up (TUG p=0.004, 10mWT p<0.001), but no significant difference in BBS was observed (p=0.132). There were no significant differences between the two groups in K-MMSE, 6MWT, and K-MBI.
This randomized control and the prospective study showed that the TSLET could independently lead to improvement in cognitive and gait function compared with conventional physical therapy in subacute stroke patients.
We hypothesized that several therapeutic elements inherent to TSLET influenced cognitive function in stroke patients. First, TSLET is a lower extremity exercise. Studies reported that physical activities like walking and jogging have a positive effect on cognitive function [21]. Research shows that lower extremity exercise has a positive effect on synaptic plasticity and connection, which seems to be mediated by a brain-derived neurotrophic factor [22-24]. Furthermore, neuroimaging research provides supporting evidence on the notion that physical activity is associated with reduced age-related atrophy of grey and white matter, and with increased grey matter density in prefrontal and temporal regions and in hippocampal volume. These results suggested that improved structural integrity of brain by exercise could lead to cognitive improvement and that the combination of exercise is more effective in neurogenesis of the brain than simple exercise [25-27]. Second, TSLET involves learning and memorization of movement pattern using visual feedback. In a similar example, dancing that requires physical activity, rhythmic motor coordination, balance, memory, sensorimotor, and visual stimulation leads to improvement in cognitive function [28] and increased hippocampal volume due to visual cue for balance [29]. Third, TSLET makes exercise more intriguing and can potentially reinforce the adherence to the training by increasing the recreational aspect of physical activity. Warburton et al. [30] showed that interactive electronic games, which require attention to objects that move across the monitor and demands weight shifting, might contribute to increased participation in physical activity.
Impaired executive function is one of the most common and crucial cognitive sequels of stroke and is present in 19% to 75% of stroke survivors [31]. Executive functions are higher-order cognitive processes that include the sequential processing, planning, and ability to concentrate. Importantly, executive functions play a significant role in determining functional recovery in stroke patients [32,33]. The digit span test is a sensitive tool for examining executive function among other cognitive functions. The digit span test forward is related to sequential processing domain. On the other hand, the digit span test backward is closely related to both sequential processing and planning domains [34]. Because K-MMSE and GDS are relatively simple tests for assessing cognitive function, they may not be sensitive enough to distinguish subtle changes in mild cognitive impairment [35].
Several studies have shown that lower extremity balance training improved gait and balance function. Kim et al. [36] reported that the TSLET showed greater improvement in BBS, 10mWT, and TUG with hemiparetic patients, and Huh et al. [9] demonstrated significant improvement in BBS and 6MWT. As expected, the results of our study proved that the TSLET group exhibited enhanced and significant improvement in BBS, TUG, and 10mWT compared with the control. However, no significant differences were observed in 6MWT and K-MBI. The BBS and TUG measures an ability to body balance while performing functional tasks like weight shifting, sitting, standing and walking. In addition, 10mWT assesses short distant gait function that depends on knee extensor strength, BBS, and TUG scores because the balance function is a major determinant of gait speed in stroke patients [37]. Also, BBS, TUG and 10mWT measures are mainly related to balance function including horizontal and vertical movement while the 6MWT measures walking endurance which mainly reflects aerobic capacity rather than balance function [38,39]. The results of 6MWT and K-MBI showed no significant difference between the TSLET group and the control group, but there was a slight improvement in the TSLET group. It is hypothesized that a longer treatment period could have led to greater improvement.
Many stroke patients have complex impairments involving cognitive, sensory, and motor functions [40]. Generally, patients receive rehabilitation therapy with a focus on a single aspect at a time. For example, physical therapy involves the enhancement of motor function and occupational therapy mainly provides training for cognitive improvement. However, many stroke patients who have complex impairment require integration of motor and cognitive function. Therefore, based on our findings that TSLET improved not only cognitive functions but also gait and balance functions, TSLET could be considered as a valuable tool in a post-stroke cognitive rehabilitation program.
However, this study has several limitations. The duration of the intervention was short and we could not study the long-term effect of TSLET. The prognosis of cognitive impairment according to the brain lesion can be variable, which we were not able to investigate in the present work. In addition, we did not exclude medication or other rehabilitative intervention like speech therapy that could affect cognitive function. Finally, we did not conduct the neurofunctional imaging studies such as fMRI and TMS that reveals cerebral plasticity. The relationship between the lesion of the stroke and the effect of TSLET, functional neuroimaging study, longer duration of the study, and the long-term effect of TSLET should be included in future studies.
In conclusion, we demonstrated that TSLET has a significant effect on cognitive and gait function compared with conventional physical therapy in patients with subacute stroke. These findings suggest that TSLET could be an effective strategy for improvement in cognitive, gait and balance functions in stroke patients.
ACKNOWLEDGMENTS
This work was supported by вҖҳSupporting Project to Test New Domestic Medical Devices in HospitalsвҖҷ funded by the Ministry of Health and Welfare and the Korea Health Industry Development Institute.
Fig.В 1.
Task-specific lower extremity training machine. (A) It consists of a balance board, a sensor, a computer system, and a screen. To prevent falling down, a harness and safety bar is used. (B) This figure shows вҖҳfruit picking gameвҖҷ. The catching cursor moves up and down according to the flexion and extension of the knee joint and moves the cursor in the left and right direction through the weight shifting.

Fig.В 2.
Flowchart represents the process of the study. Three of 38 patients dropped out, 1 from the TSLET group, and 2 from the control group due to follow-up loss. TSLET, task-specific lower extremity training.
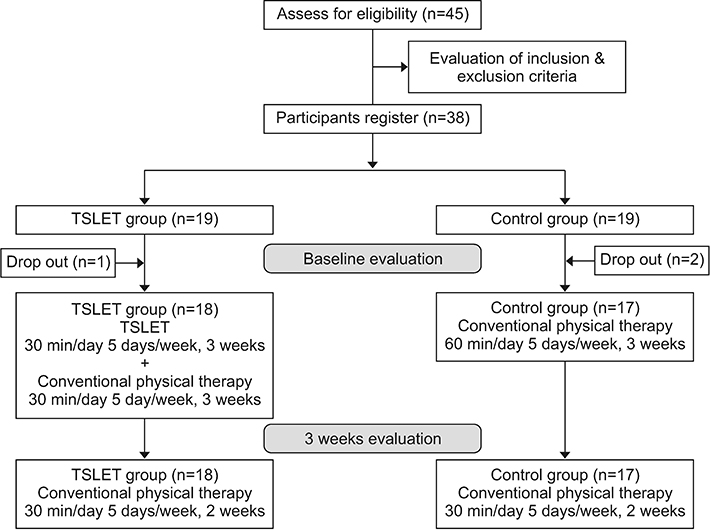
Fig.В 3.
Changes in parameters of the primary outcome (digit span test). (A) VST-F, (B) VST-B, (C) AST-F, and (D) AST-B. In the TSLET group, a significant time versus group interaction was observed for VST-B improvement (p=0.031). TSLET, task-specific lower extremity training; VST-F, visual digit span test forward; VST-B, visual digit span test backward; AST-F, auditory digit span test forward; AST-B, auditory digit span test backward.

TableВ 1.
Demographic and clinical characteristics of the subjects
TableВ 2.
Changes in parameters of secondary outcomes for cognitive, gait, balance, and functional ability
| Parameter | Group | Time 1 (baseline) | Time 2 (3 weeks) | Time 3 (5 weeks) |
|---|---|---|---|---|
| K-MMSE | TSLET | 23.89Вұ4.04 | 25.44Вұ4.18 | 26.23Вұ3.72 |
| Control | 23.17Вұ3.91 | 23.88Вұ3.93 | 24.12Вұ3.94 | |
| GDS | TSLET | 3.42Вұ1.02 | 2.61Вұ1.10* | 2.47Вұ1.12* |
| Control | 3.23Вұ1.03 | 2.94Вұ0.90 | 2.76Вұ0.83 | |
| BBS | TSLET | 31.42Вұ9.14 | 39.19Вұ9.12* | 43.64Вұ7.98* |
| Control | 31.94Вұ7.52 | 34.94Вұ8.84 | 36.59Вұ9.57 | |
| TUG | TSLET | 27.63Вұ17.61 | 25.13Вұ17.20* | 22.47Вұ16.30* |
| Control | 27.93Вұ17.66 | 25.47Вұ16.70 | 26.88Вұ18.75 | |
| 10mWT | TSLET | 23.86Вұ12.83 | 21.11Вұ12.06* | 18.65Вұ14.50* |
| Control | 19.33Вұ12.83 | 18.69Вұ11.03 | 21.34Вұ13.22 | |
| 6MWT | TSLET | 225.10Вұ85.70 | 223.66Вұ105.77 | 268.19Вұ124.18 |
| Control | 220.75Вұ79.81 | 229.12Вұ99.75 | 229.14Вұ97.90 | |
| K-MBI | TSLET | 56.47Вұ21.46 | 66.67Вұ18.21 | 72.88Вұ20.00 |
| Control | 55.00Вұ20.07 | 60.59Вұ16.95 | 62.06Вұ16.71 |
Values are presented as meanВұstandard deviation.
TSLET, task-specific lower extremity training; K-MMSE, Korean version of Mini-Mental State Examination; GDS, Global Deterioration Scale; BBS, Berg Balance Scale; TUG, Timed Up and Go Test; 10mWT, 10 meters Walking Test; 6MWT, 6 minutes Walking Test; K-MBI, Korean version of Modified Barthel index.
REFERENCES
1. Tatemichi TK, Desmond DW, Stern Y, Paik M, Sano M, Bagiella E. Cognitive impairment after stroke: frequency, patterns, and relationship to functional abilities. J Neurol Neurosurg Psychiatry 1994;57:202-7.



2. Jin YP, Di Legge S, Ostbye T, Feightner JW, Hachinski V. The reciprocal risks of stroke and cognitive impairment in an elderly population. Alzheimers Dement 2006;2:171-8.


3. Mok VC, Wong A, Lam WW, Fan YH, Tang WK, Kwok T, et al. Cognitive impairment and functional outcome after stroke associated with small vessel disease. J Neurol Neurosurg Psychiatry 2004;75:560-6.



4. Tatemichi TK, Paik M, Bagiella E, Desmond DW, Pirro M, Hanzawa LK. Dementia after stroke is a predictor of long-term survival. Stroke 1994;25:1915-9.


5. Haggard P, Cockburn J, Cock J, Fordham C, Wade D. Interference between gait and cognitive tasks in a rehabilitating neurological population. J Neurol Neurosurg Psychiatry 2000;69:479-86.



6. El-Tamawy MS, Abd-Allah F, Ahmed SM, Darwish MH, Khalifa HA. Aerobic exercises enhance cognitive functions and brain derived neurotrophic factor in ischemic stroke patients. NeuroRehabilitation 2014;34:209-13.


7. Fernandez-Gonzalo R, Fernandez-Gonzalo S, Turon M, Prieto C, Tesch PA, Garcia-Carreira Mdel C. Muscle, functional and cognitive adaptations after flywheel resistance training in stroke patients: a pilot randomized controlled trial. J Neuroeng Rehabil 2016;13:37.



8. Wayne PM, Walsh JN, Taylor-Piliae RE, Wells RE, Papp KV, Donovan NJ, et al. Effect of tai chi on cognitive performance in older adults: systematic review and meta-analysis. J Am Geriatr Soc 2014;62:25-39.



9. Huh JS, Lee YS, Kim CH, Min YS, Kang MG, Jung TD. Effects of balance control training on functional outcomes in subacute hemiparetic stroke patients. Ann Rehabil Med 2015;39:995-1001.



10. Hung JW, Chou CX, Chang HF, Wu WC, Hsieh YW, Chen PC, et al. Cognitive effects of weight-shifting controlled exergames in patients with chronic stroke: a pilot randomized comparison trial. Eur J Phys Rehabil Med 2017;53:694-702.


11. Kim YH, Shin SH, Park SH, Ko MH. Cognitive assessment for patient with brain injury by computerized neuropsychological test. J Korean Acad Rehabil Med 2001;25:209-16.
12. Kool W, Conway AR, Turk-Browne NB. Sequential dynamics in visual short-term memory. Atten Percept Psychophys 2014;76:1885-901.



13. Hirayama S, Terasawa K, Rabeler R, Hirayama T, Inoue T, Tatsumi Y, et al. The effect of phosphatidylserine administration on memory and symptoms of attention-deficit hyperactivity disorder: a randomised, double-blind, placebo-controlled clinical trial. J Hum Nutr Diet 2014;27 Suppl 2:284-91.


14. Oh E, Kang Y, Shin JH, Yeon BK. A validity study of KMMSE as a screening test for dementia: comparison against a comprehensive neuropsychological evaluation. Dement Neurocognitive Disord 2010;9:8-12.
15. Kang Y, Na DL, Hahn S. A validity study on the Korean Mini-Mental State Examination (K-MMSE) in dementia patients. J Korean Neurol Assoc 1997;15:300-8.
16. Eisdorfer C, Cohen D, Paveza GJ, Ashford JW, Luchins DJ, Gorelick PB, et al. An empirical evaluation of the Global Deterioration Scale for staging AlzheimerвҖҷs disease. Am J Psychiatry 1992;149:190-4.


17. Blum L, Korner-Bitensky N. Usefulness of the Berg Balance Scale in stroke rehabilitation: a systematic review. Phys Ther 2008;88:559-66.



18. Yelnik A, Bonan I. Clinical tools for assessing balance disorders. Neurophysiol Clin 2008;38:439-45.


19. Pohl PS, Duncan PW, Perera S, Liu W, Lai SM, Studenski S, et al. Influence of stroke-related impairments on performance in 6-minute walk test. J Rehabil Res Dev 2002;39:439-44.

20. Jung HY, Park BK, Shin HS, Kang YK, Pyun SB, Paik NJ, et al. Development of the Korean version of Modified Barthel Index (K-MBI): multi-center study for subjects with stroke. J Korean Acad Rehabil Med 2007;31:283-97.
21. Bramell-Risberg E, Jarnlo GB, Elmstahl S. Separate physical tests of lower extremities and postural control are associated with cognitive impairment: results from the general population study Good Aging in Skane (GAS-SNAC). Clin Interv Aging 2012;7:195-205.



22. Cotman CW, Berchtold NC, Christie LA. Exercise builds brain health: key roles of growth factor cascades and inflammation. Trends Neurosci 2007;30:464-72.


23. Gomez-Pinilla F, Vaynman S, Ying Z. Brain-derived neurotrophic factor functions as a metabotrophin to mediate the effects of exercise on cognition. Eur J Neurosci 2008;28:2278-87.



24. Vaynman S, Ying Z, Gomez-Pinilla F. Hippocampal BDNF mediates the efficacy of exercise on synaptic plasticity and cognition. Eur J Neurosci 2004;20:2580-90.


25. Colcombe SJ, Erickson KI, Raz N, Webb AG, Cohen NJ, McAuley E, et al. Aerobic fitness reduces brain tissue loss in aging humans. J Gerontol A Biol Sci Med Sci 2003;58:176-80.



26. Gordon BA, Rykhlevskaia EI, Brumback CR, Lee Y, Elavsky S, Konopack JF, et al. Neuroanatomical correlates of aging, cardiopulmonary fitness level, and education. Psychophysiology 2008;45:825-38.



27. Erickson KI, Prakash RS, Voss MW, Chaddock L, Hu L, Morris KS, et al. Aerobic fitness is associated with hippocampal volume in elderly humans. Hippocampus 2009;19:1030-9.



28. Kattenstroth JC, Kolankowska I, Kalisch T, Dinse HR. Superior sensory, motor, and cognitive performance in elderly individuals with multi-year dancing activities. Front Aging Neurosci 2010;2:31.



29. Hufner K, Binetti C, Hamilton DA, Stephan T, Flanagin VL, Linn J, et al. Structural and functional plasticity of the hippocampal formation in professional dancers and slackliners. Hippocampus 2011;21:855-65.


30. Warburton DE, Bredin SS, Horita LT, Zbogar D, Scott JM, Esch BT, et al. The health benefits of interactive video game exercise. Appl Physiol Nutr Metab 2007;32:655-63.


31. Barker-Collo S, Feigin V. The impact of neuropsychological deficits on functional stroke outcomes. Neuropsychol Rev 2006;16:53-64.


32. Liu-Ambrose T, Pang MY, Eng JJ. Executive function is independently associated with performances of balance and mobility in community-dwelling older adults after mild stroke: implications for falls prevention. Cerebrovasc Dis 2007;23:203-10.


33. Lesniak M, Bak T, Czepiel W, Seniow J, Czlonkowska A. Frequency and prognostic value of cognitive disorders in stroke patients. Dement Geriatr Cogn Disord 2008;26:356-63.


34. Schofield NJ, Ashman AF. The relationship between digit span and cognitive processing across ability groups. Intelligence 1986;10:59-73.

35. Tombaugh TN, McIntyre NJ. The mini-mental state examination: a comprehensive review. J Am Geriatr Soc 1992;40:922-35.


36. Kim CH, Byun SD, Shin OS, Kim TG, Kwon SM, Noh JH, et al. Effect of the balance control of the affected lower extremity on balance and gait in hemiparetic patients. J Korean Acad Rehabil Med 2008;32:394-9.
37. Hyun CW, Han EY, Im SH, Choi JC, Kim BR, Yoon HM, et al. Hemiparetic knee extensor strength and balance function are predictors of ambulatory function in subacute stroke patients. Ann Rehabil Med 2015;39:577-85.



38. Graham JE, Ostir GV, Fisher SR, Ottenbacher KJ. Assessing walking speed in clinical research: a systematic review. J Eval Clin Pract 2008;14:552-62.










