Changes in Function and Muscle Strength of Encephalitis Survivors After Inpatient Rehabilitation
Article information
Abstract
Objective
To investigate the clinical demographics and rehabilitative assessments of encephalitis survivors admitted to a rehabilitation center, and to confirm the effects of inpatient rehabilitation manifested by changes in muscle strength and function after hospitalization.
Methods
Data of encephalitis survivors who received rehabilitation at our institution from August 2009 to August 2019 were reviewed. Medical charts were retrospectively reviewed, and motor, functional, and cognitive assessments were collected. Manual muscle testing (MMT), Fugl-Meyer Assessment (FMA), Berg Balance Scale (BBS), Functional Ambulation Category (FAC), Korean version of Modified Barthel Index (K-MBI), grip strength, Box and Block Test (BBT), and Korean version of Mini-Mental State Examination (K-MMSE) were performed, and the results upon admission and discharge were compared and analyzed.
Results
Most of the patients with encephalitis admitted to our institution had viral or autoimmune etiologies. The assessment results of 18 encephalitis patients upon admission and discharge were compared. The total K-MBI score, FAC, grip strength, and BBT significantly improved, but not the MMT and FMA. Subgroup analysis was performed for viral and autoimmune encephalitis, which are the main causes of the disease, but there was no difference in items with significant changes before and after hospitalization.
Conclusion
Encephalitis survivors showed a significant improvement in functional assessment scale during their hospital stay through rehabilitation, without significant changes in motor strength. Hence, we can conclude that encephalitis survivors benefit from inpatient rehabilitation, targeting functional gains in activities of daily living training more than motor strength.
INTRODUCTION
Encephalitis can be classified as infectious, immunemediated, or unknown, depending on its etiology [1]. Although various factors, such as viruses, bacteria, fungi, and parasites, may cause infectious encephalitis, viruses are the most prevalent cause [1,2]. Immune-mediated causes include autoimmune encephalitis, such as N-methyl-D-aspartate (NMDA) receptor or voltage-gated potassium channel antibody type, as well as secondary causes due to systemic vasculitis, multiple sclerosis, and tumorous conditions [1]. Among the patients diagnosed with encephalitis, cases with undetermined etiology were classified as unknown. According to articles published in the United Kingdom and United States, the most common etiology was infectious, followed by unknown [1,2]. Since patients with encephalitis may have different symptoms and prognoses depending on the cause [1], it is important for clinicians treating these patients to be aware of the etiology.
The reported global incidence of encephalitis varies between 3.5 and 7.4 per 100,000 person-years [3]. Despite its low incidence, encephalitis survivors suffer from neurological sequelae for a long period of time. Thus, the medical and social burden of encephalitis to individuals and the society are significant. Encephalitis also has a comparable burden on medical expenses to that of traumatic brain injury (TBI) or stroke [4].
Patients who recover from the acute stage of encephalitis might achieve complete functional recovery, but persistent and severe functional decline occurred in some patients, which became an obstacle in living adjustments and return to society. For example, 20%–60% of those who recovered from herpes simplex encephalitis reported chronic sequelae, including neurological abnormalities, movement disorders, aphasia, abnormal behavior, and cognitive decline [5]. Therefore, rehabilitative intervention for encephalitis survivors is necessary to minimize chronic sequelae, including drug treatment and physical and behavioral rehabilitation [6].
Functional improvement in encephalitis survivors under rehabilitative intervention has been shown in several case reports and case series [4]. The UK guidelines on encephalitis express that brain injury rehabilitation with a specialist aids in recovery [7,8]. Despite these guidelines, there are only a few studies on the rehabilitation of patients with encephalitis.
As discussed, despite encephalitis survivors experiencing sequelae for a long period of time and requiring rehabilitation, which consequently incurs high medical costs, there are few studies on the effectiveness of rehabilitation treatment. We believe that research planning and investigation is challenging due the low incidence of encephalitis. Therefore, in this study, we aimed to investigate the clinical characteristics and motor and functional deficits of encephalitis survivors admitted to a rehabilitation center, and to evaluate the effects of inpatient rehabilitation manifested by changes in muscle strength and function after hospitalization.
MATERIALS AND METHODS
This study prepared a retrospective chart review of patients with encephalitis who were treated at National Rehabilitation Center (NRC) from August 2009 to August 2019. As most patients at NRC had already been diagnosed, initial evaluation and diagnosis during the acute stage could not be performed. Thus, we adopted the diagnosis of medical records made by the admitting medical centers. In July 2019, we obtained approval from the Institutional Review Board of the National Rehabilitation Center to undertake this study (No. NRC-2019-04-030). As this study involved a retrospective design with anonymized data, a requirement for informed patient consent was waived.
Patients aged 18–65 years who were diagnosed with encephalitis between 1 and 24 months before hospitalization were included. Only patients hospitalized for more than a month were included in the study. Patients with meningitis, meningoencephalitis, encephalomyelitis, and limited physical activity due to severe neuropsychiatric, medical, or orthopedic conditions were excluded.
As a routine rehabilitation treatment protocol at our center, basic physical therapy (PT) and occupational therapy (OT) are implemented. For patients who have passed 3 months or more since the disease onset, PT for 30 minutes twice, OT for 30 minutes once, and exercise using machines such as an ergometer for 30 minutes twice, are conducted in a day. PT comprises a range of motion exercises, including muscle strengthening, stretching, gait training, and aerobic exercises, while OT comprises training for hand function, training for activities of daily living (ADL), and sensory integration therapy.
Based on the chart reviews, the patients’ diagnoses were initially classified into infectious and immunemediated causes; if the exact cause was unspecified, encephalitis was classified as unknown. However, as a result of the investigation, all infectious causes were identified as viruses, and all immune-mediated causes were due to autoimmunity. Hence, these were termed viral and autoimmune encephalitis, respectively. In addition to the general characteristics, such as sex and age, neurologic deficits, including the type of paralysis and seizure history, possibly affecting the patient’s function, were also investigated. We defined quadriplegia as neurological deficits in both upper and lower limbs, hemiplegia as neurological deficits in the left or right upper and lower limbs, and diplegia as neurological deficits in both lower limbs. Seizure history included any type of seizure or status epilepticus experienced at our center or other medical centers. Data on motor, functional, and cognitive assessments, which evaluated muscle strength, motor function, ADL, and other features, were investigated from the perspective of rehabilitation medicine. Among these results, we analyzed and compared the assessments before and after inpatient rehabilitation.
The routine evaluation items of our center have progressed over the past few years. When designing and conducting this study, all items were routinely evaluated. However, data might be missing for patients hospitalized before these routine evaluation items were established. If the patient’s results of each assessment performed upon hospitalization and discharge were missing, statistical analysis was performed without the patient’s result.
In this study, we focused on the changes in muscle strength and function; hence, we collected data using the following strategy. The results of manual muscle testing (MMT) were collected to investigate the patient’s muscle strength, which was indicated as a Medical Research Council (MRC) grade of 5 points. Shoulder and hip flexion were selected for the upper and lower extremities, respectively, since shoulder joint movement is essential for the ADL [9], and the hip flexor is a key component muscle for walking with an essential MMT grade ≥3 to be able to walk outdoors [10]. To investigate the functional assessments of patients with encephalitis, several assessment tools were used, such as the Fugl-Meyer Assessment (FMA), reflecting the overall motor function; Berg Balance Scale (BBS), evaluating balance ability; Functional Ambulation Category (FAC), evaluating gait function; Korean version of Modified Barthel Index (K-MBI), reflecting information on ADL; and dynamometry and Box and Block Test (BBT), investigating grip strength, since deterioration of hand function and grip strength adversely affects the performance of ADL and consequently worsens overall function [11,12]. However, in the case of grip strength, normal ranges vary according to sex and age, possibly generating bias and errors if we simply compared the average grip strength. Therefore, the grip strength results, regardless of age, were classified as “normal” or “abnormal,” which was defined as <27 kg and <16 kg in men and women, respectively [12]. The results of the Korean version of Mini-Mental State Examination (K-MMSE), which was conducted to screen cognitive deficits, were also investigated and included.
Data were analyzed using IBM SPSS version 26 (IBM, Armonk, NY, USA). Descriptive statistical analysis was performed for all the items examined, aiming to determine whether the general demographic features of the population differed according to the disease etiology. Fisher exact test was used to verify whether the basic demographic features and disease characteristics showed differences between the viral and autoimmune groups, excluding the group with unknown causes. Age was compared using the Kruskal-Wallis test. The Wilcoxon ranksum test was used to compare changes in assessment after rehabilitation between the two groups. McNemar test was used to analyze changes in grip strength after inpatient rehabilitation. Statistical significance was set at p<0.05.
RESULTS
A total of 23 patients were admitted for encephalitis due to various causes and received inpatient rehabilitation. After excluding three patients because of age and two patients who dropped out because of other medical conditions, a total of 20 patients were enrolled in the study, and their medical charts were reviewed (Fig. 1).

Flow chart for the inclusion and/or exclusion of study participants. NRC, National Rehabilitation Center.
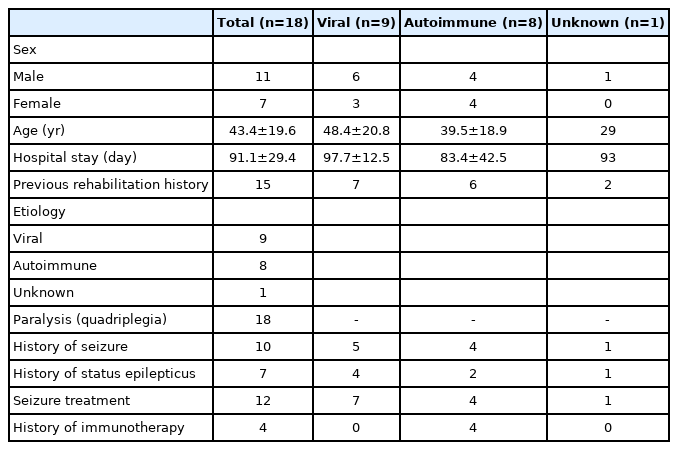
Table 1 summarizes the basic demographic and disease characteristics of encephalitis survivors and presents the results based on their etiologies, which are viral, autoimmune, and unknown. Among the 18 patients, 11 were men. The mean disease onset duration before admission was 211 days, and the mean hospital stay was 91.1 days. Nine, eight, and one patient had viral, autoimmune, and unknown etiologies, respectively. The patient with unknown etiology was included in the total encephalitis patient analysis, but excluded from the subgroup analysis. All 18 patients showed quadriplegia, and 10 (five, four, and one with viral, autoimmune, and unknown etiologies, respectively) experienced seizures, of which, seven (four, two, and one with viral, autoimmune, and unknown etiologies, respectively) were diagnosed with status epilepticus. Among those with autoimmune encephalitis, four patients had a history of immunotherapy.
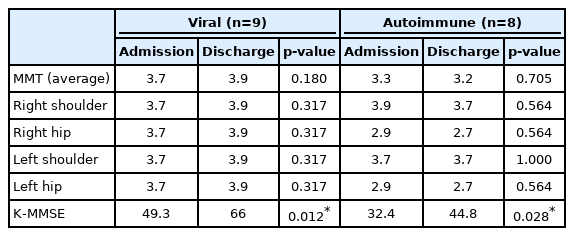
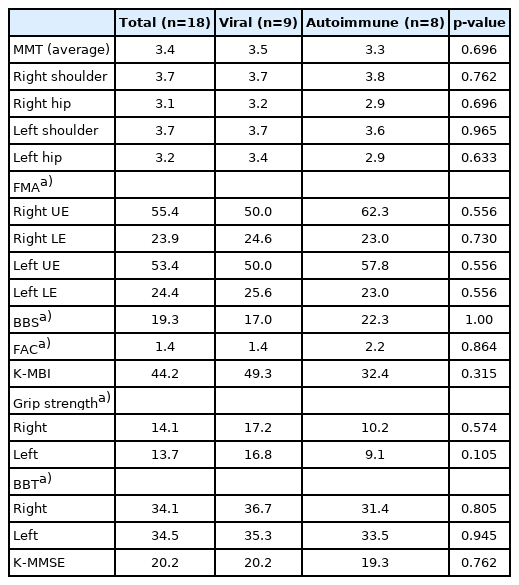
Table 2 shows the collected results of motor and functional assessments per etiology at the time of admission, and the differences between the groups were verified and presented as p-values. The average MMT grade was 3.4 in the MRC grade. The average right and left FMA and BBS scores collected from nine patients were 79.3, 77.9, and 19.3, respectively. The average K-MBI score of all 18 patients was 44.2 points. The average right and left grip strength of 17 patients were 14.1 and 13.7 kg, respectively. The average right and left BBT scores of 13 patients were 34.1 and 34.5, respectively. The average FAC scale score of 15 patients was 1.9. The average K-MMSE score of all 18 patients was 20.2. No significant differences were observed between the viral and autoimmune encephalitis groups.
Comparison of motor power and functional assessments of encephalitis survivors at admission between viral and autoimmune etiologies
Table 3 shows the differences between the assessments at the time of admission and the assessments performed before discharge among the encephalitis survivors. There were no significant changes in MMT and FMA; however, significant improvements were observed in the BBS score (from 19.3 at admission to 31.8 at discharge), total K-MBI score (from 44.2 to 58.3), FAC scale (from 1.9 to 2.9), bilateral grip strength (right side, from 14.0 to 22.0; left side, from 13.8 to 20.9), and BBT score (right side, from 39.6 to 46.5; left side, from 39.9 to 50.1). The K-MBI scores are presented in Supplementary Table S1. The grip strengths were classified as either normal or abnormal according to the reference values by Dodds et al. [12], and abnormal to normal changes after inpatient rehabilitation were analyzed accordingly. In terms of bilateral grip strength, four of the 11 abnormal patients showed improvement to normal ranges, but the results were not statistically significant.
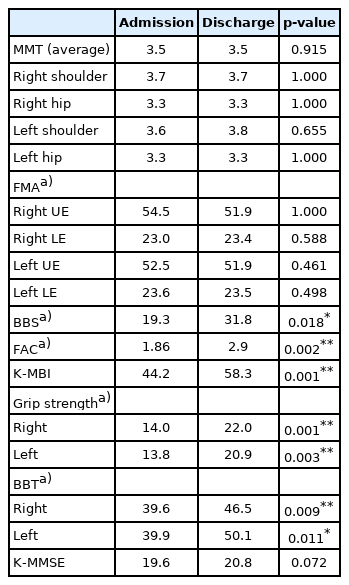
Comparison of changes in motor power and functional assessments after inpatient rehabilitation for encephalitis survivors
In Table 4, patients are classified into viral and autoimmune encephalitis groups, and changes in MMT and K-MBI scores before and after hospitalization are presented with corresponding p-values. For both groups, changes in MMT were not statistically significant, whereas improvements in K-MBI scores were significant.
DISCUSSION
According toa previous meta-analysis on the rehabilitation of patients with encephalitis, most studies have used the Functional Independence Measure (FIM) or cognitive assessment measures [4]. Previous studies that used various rehabilitative assessments were clinical series with a small sample size [13]. Our study aimed to overcome the limitations of previous studies and present more convincing evidence of rehabilitation for patients with encephalitis using a variable assessment tool for motor power and function. We included detailed rehabilitation measures, such as MMT, FMA, BBS, FAC, ADL, grip strength, and BBT, using a sample size of 18, which was larger than that reported in previous studies. We aimed to determine its clinical significance among patients with encephalitis and the differences in rehabilitation effects in those patients compared to patients with other brain injuries, such as stroke. As such, we determined that inpatient rehabilitation caused functional improvement in encephalitis survivors, even without significant changes in motor strength. According to Bohannon [14], strength in stroke patients is associated with the ability to perform functional tasks, and that most stroke patients reach the best neurological level within 11 weeks of onset, and only show minor measurable improvement 6 months after onset [15,16]. Although the encephalitis survivors in this study were chronic patients who had already passed 211 days after the onset on average, they showed significant functional changes. Considering this, it would be reasonable to greatly consider functional improvement during rehabilitation for encephalitis survivors.
We collected data from 18 patients and investigated their basic demographic and clinical features. As a set number of rehabilitative treatments were provided according to policy, we assumed that patients with encephalitis were exposed to a relatively similar treatment environment. All 18 patients had chronic encephalitis upon hospital admission, approximately 211 days from disease onset. The length of stay did not differ significantly between the viral, autoimmune, and unknown groups; therefore, we inferred that patients enrolled in the study would have received a relatively similar dosage of rehabilitation treatment.
Patients with encephalitis showed significant functional changes following inpatient rehabilitation, despite their chronic presentation. Recovery of encephalitis survivors in the rehabilitative aspect was relatively slower than that of stroke or TBI patients [17], which is consistent with our results that show how effective rehabilitation is even in patients with chronic encephalitis. In this context, patients with encephalitis would have a more prolonged therapeutic window in functional recovery than patients of stroke or TBI; thus, rehabilitative treatment should be performed for a longer period of time for these patients.
Seizures are known to be an important factor affecting the prognosis of both infectious and autoimmune encephalitis, with the latter considered an important clinical cause of chronic epileptogenesis [18-20]. Thus, we predicted that seizure-related features would be more frequent in autoimmune encephalitis, resulting in a worse prognosis. In this study, however, the proportion of patients who experienced seizures was similar in both viral (56%) and autoimmune (50%) encephalitis, and the proportion of status epilepticus appeared to be higher due to viral (80%) etiologies than autoimmune (50%). Although there was no statistically significant difference, the anti-epileptic effect of immunotherapy was expected to play a major role in this result. Since autoimmune antibodies against neuronal cell-surface proteins are involved in the epileptogenic mechanism of autoimmune encephalitis [20], such mechanism would be inhibited by appropriate immunotherapy. Awareness of encephalitis-related seizure features would provide information for adequate monitoring and control, enabling rehabilitation experts to predict their prognosis.
All patients showed quadriplegia, with no significant differences in basic demographic features or clinical characteristics, motor, functional, and cognitive assessments at the time of admission according to etiology. Based on these results, it could be assumed that various etiologies share similar clinical features in terms of functional and rehabilitative outcome measures.
In this study, there was no significant change in FMA or MMT after inpatient rehabilitation treatment, but the BBS, K-MBI, FAC, grip strength, and BBT scores significantly improved. We assumed that the significant improvement in BBS without any significant change in FMA or MMT was due to posture or coordination improvement through rehabilitation, rather than a recovery in muscle strength. K-MBI showed significant improvement not only in the total scores, but also in individual items, except for bowel and bladder control (Supplementary Table S1). These findings were consistent with those of a previous study showing that inpatient rehabilitation for encephalitis improves functional outcomes [17].
Hand function is directly related to the patient’s ADL, and this study included grip strength and BBT as indicators of hand function [11]. Significant improvements in these indicators elucidated the functional improvement of ADL, suggesting that it can be used to measure rehabilitative outcomes in patients with encephalitis. A previous case series showed that two out of three patients had improved hand function using the Nine-Hole Peg Test [13]. Regarding grip strength, varied normal ranges according to sex and age deemed it unreasonable to confirm the effect of rehabilitation by merely comparing the average grip strength values, as shown in Table 3. To compensate, we introduced “normal” and “abnormal” categories according to sex [12] and examined how many patients with abnormal hand grip strength showed improvement to normal values, which did not show statistical significance, with a p-value of 0.063.
To date, many studies on encephalitis rehabilitation have focused on specific etiologies. In contrast, our study attempted to assess the rehabilitation of encephalitis survivors more comprehensively. As mentioned, significant changes in functional assessment were confirmed in all patients with encephalitis, including those with viral, autoimmune, and unknown etiologies, without changes in muscle strength. We performed a subgroup analysis for the two subgroups, and the results are shown in Table 4. Functional improvements in ADL without muscle strength changes observed in the total encephalitis patient group were confirmed in the same manner within the subgroup analysis for the viral and autoimmune groups. Based on our results, we deduced a general treatment idea that the goal of rehabilitation should be focused on functional gain for patients with encephalitis, regardless of etiologies.
This study has some limitations, one of which is the small sample size of 18. Most of the limitations of previous studies related to encephalitis rehabilitation were the small sample size and the case report (series) design [4]. Nevertheless, the study with the largest sample size before ours was 13, with patients aged 5–14 years [21]. Moorthi et al. [17] analyzed a sample size of eight patients aged 5–75 years. In this context, our study had a larger sample size than that of previous studies. However, there was still a limitation in statistical analysis due to the small sample size. To prove that inpatient rehabilitation contributed to the improvement of the functions of encephalitis survivors, it was necessary to control the confounding variables that could affect the functional change and implement a suitable method of statistical analysis. However the most suitable method was limited due to the small sample size; therefore, we confirmed the effect of inpatient rehabilitation treatment indirectly by simply comparing the functions at the time of admission and discharge.
Another limitation is the insufficient data in the cognitive domain. Cognitive deficits are a common clinical manifestation of encephalitis, to which rehabilitative interventions are effective [22-26]. However, in this study, only the K-MMSE was used to evaluate cognitive function due to incomplete records of various cognitive assessment tools. We believe that with a standardized cognitive assessment tool, more significant rehabilitation effects can be demonstrated in future studies.
Despite these limitations, this study observed positive changes in several functional scales without changes in MMT, including motor function and ADL. Therefore, we could draw a distinction between encephalitis and other acquired brain injuries, such as stroke, which usually show simultaneous muscle strength and functional improvements. Hence, it is important to prioritize functional gain during the rehabilitation of patients with encephalitis, leading to more individualized and tailored management. To compensate for the aforementioned limitations, future studies should include various functional and standardized cognitive assessments to thoroughly understand the effects of rehabilitation on encephalitis survivors.
Notes
No potential conflict of interest relevant to this article was reported.
Conceptualization: Cheong IY. Methodology: Cheong IY. Formal analysis: Cheong IY, Kim T. Project administration: Cheong IY, Kim T. Writing – original draft: Kim T. Writing – review and editing: Cheong IY, Kim T. Approval of final manuscript: all authors.
SUPPLEMENTARY MATERIALS
Supplementary materials can be found via http://doi.org/10.5535/arm.21133.
Average score of each K-MBI items (n=18)