Effect of Extracorporeal Shockwave Therapy Versus Intra-articular Injections of Hyaluronic Acid for the Treatment of Knee Osteoarthritis
Article information
Abstract
Objective
To evaluate and compare the effects and outcomes of extracorporeal shock wave therapy (ESWT) and intra-articular injections of hyaluronic acid (HA) in patients with knee osteoarthritis (OA).
Methods
Of the 78 patients recruited for the study, 61 patients met the inclusion criteria. The enrolled patients were randomly divided into two groups: the ESWT group and the HA group. The ESWT group underwent 3 sessions of 1,000 shockwave pulses performed on the affected knee with the dosage adjusted to 0.05 mJ/mm2 energy. The HA group was administered intra-articular HA once a week for 3 weeks with a 1-week interval between each treatment. The results were measured with the visual analogue scale (VAS), Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC), Lequesne index, 40-m fast-paced walk test, and stair-climb test (SCT). A baseline for each test was measured before treatment and then the effects of the treatments were measured by each test at 1 and 3 months after treatment.
Results
In both groups, the scores of the VAS, WOMAC, Lequesne index, 40-m fast-paced walk test, and SCT were significantly improved in a time-dependent manner (p<0.01). There were no statistically significant differences measured at 1 and 3 months after treatment between the two groups (p>0.05).
Conclusion
The ESWT can be an alternative treatment to reduce pain and improve physical functions in patients with knee OA.
INTRODUCTION
Osteoarthritis (OA), a progressive degenerative joint disease, is associated with a variety of risk factors such as age, heredity, obesity, and external injury. OA occurs in varied foci that range from the cartilages to entire joints. The pathologic processes affect entire joints such as the synovium, bone, and cartilage, which results in degeneration of the joint. OA is characterized by the degeneration of the articular cartilage, osteophyte formation, subchondral bone sclerosis, degeneration of meniscus, and synovial proliferation. Such pathologic features cause pain, articular dysfunction, disability, and degrade quality of life. Most treatment goals of OA involve pain reduction, improvement of joint mobility, treatment of dysfunction, and improvement in the quality of life [1].
Knee joint arthritis is the most common disease caused by OA. Various treatment options are available for knee OA. The ideal treatment is a combined treatment of non-pharmacological and pharmacological therapies and in cases where symptomatic treatment is ineffective, the patient can resort to surgery. The non-pharmacological treatments include muscle strengthening, exercise, weight management, thermal modalities, and transcutaneous electrical nerve stimulation (TENS). The pharmacological treatments include acetaminophen, non-steroidal anti-inflammatory drugs (NSAIDs), capsaicin, opioids, and intra-articular injections of corticosteroids or hyaluronic acid (HA) [2]. Most of the patients with knee OA are elderly with comorbidities [3]. In this context, the intra-articular therapies were widely employed. The intra-articular injection of HA and corticosteroids is a recommended treatment for knee OA [4]. However, although diverse treatments are available for knee OA, the pain often persists [5]. As such, research for new knee OA treatments remains of interest.
Extracorporeal shock wave therapy (ESWT) is widely used for treating muscular skeletal diseases and pain relief. The curative effects of ESWT have been proved in cases involving plantar fasciitis, calcific tendinitis of the shoulder, epicondylitis, patellar tendinopathy, achilles tendinopathy, non-union and delayed union of long bone fracture, and avascular necrosis of the femoral head. Hence, ESWT is now extensively used for such afflictions [6]. Recently, ESWT was applied for knee OA by a veterinarian who had introduced ESWT for treating knee OA in horses [78]. The curative effects of ESWT for knee OA are reported in some animal studies. These reports reveal that the application of ESWT to the arthritic joint of animals delayed the OA progression, improved motor dysfunction, reduced pain, and observed a regression of OA and chondroprotective effects [9101112]. Some recent studies elucidated that ESWT has an effect on pain relief and improvement of knee functions when applied to human subjects [131415]. Considering these study results, ESWT can be a beneficial option for knee OA treatment.
A previous study comparing the effects of ESWT and intra-articular corticosteroids injection for knee OA has been reported [16]. In the study, pain relief, improved dysfunction, and improved range of motion of knee joints were predominantly observed in the ESWT and intraarticular corticosteroids injection group as compared to the control group. Also, these effects were detected more in the ESWT group than in the intra-articular corticosteroids injection group. However, a direct comparison of ESWT and intra-articular HA injection for the treatment for symptomatic human knee OA has rarely been investigated.
This study therefore compared the effects of ESWT and intra-articular HA injection in patients with knee OA.
MATERIALS AND METHODS
Participants
Patients suffering from knee pain who visited a general hospital for a minimum of 3 months were recruited from January 2015 to December 2015. The inclusion criteria were as follows: (1) people aged 45 years or more; (2) subjects diagnosed with knee OA according to the clinical criteria of the American College of Rheumatology [17]; (3) subjects diagnosed with grade II or III OA during radiological examination as defined by the radiological classification of Kellgren and Lawrence (K-L) scale for knee OA [18]; (4) subjects with tenderness in the medial tibial plateau area; and (5) subjects who had pain on one side of the knee. In cases where patients experienced pain in both knees, we applied ESWT and HA injections on the more painful side. The exclusion criteria included: (1) subjects having other illnesses of neurologic diseases, cardiac disorders, hemodynamically unstable systems, or physical functions; (2) subjects who have received intra-articular injection treatment or ESWT on the affected knee within the past 6 months; (3) subjects with previous medical histories involving the affected knee such as external injuries, surgeries, cancer, and malignant tumors; (4) subjects who did not permit radiological examinations or magnetic resonance images (MRIs).
Of the 78 subjects recruited, 61 patients met the inclusion criteria. This research was approved by the Institutional Review Board of the Gwangju Veterans Hospital (2015-1-1) and carried out at a single medical center. Consent forms were received before commencement of the study. This study was conducted in compliance with the principles set forth in the Declaration of Helsinki.
Methods
A prospective randomized controlled study was carried out. Computer aided randomization was performed before conducting the study. Independent investigators not involved in treating the patients or measuring their outcomes divided the patients into the ESWT group (n=31; receiving ESWT) or the HA group (n=30; receiving intraarticular injection of HA).
For the ESWT group, we employed the Dornier EPOS Ultra device (Dornier MedTech, Kennesaw, GA, USA) that was approved in 2001. The ESWT has an electromagnetic source and uses a smart focus type. A total of 3 sessions of ESWT were performed weekly for the 31 patients in the ESWT group. During ESWT, the patients had to lie on the table in a supine position with their knees bent to about 90°. At every treatment session, 1,000 pulses of shockwave with an energy dose of 0.05 mJ/mm2 were applied to the tender point of the medial tibial plateau area in the affected knee [15]. ESWT was performed by physicians who did not participate in randomizing the patients and measuring their outcomes.
In the HA group, 2 mL of HA (sodium hyaluronate, 20 mg/2 mL, molecular weight 3×106 Daltons) was administered weekly for 3 consecutive weeks into the affected knee using a lateral midpatellar approach (Fig. 1). Patients had to lie on the table in a supine position with their knees extended during the intra-articular HA injection. The medicinal substances were administered by transversely inserting a needle between the articular surface and the patellofemoral joint in the midpoint of the patella, after pushing the patella upward and shifting it to the lateral side [19]. All procedures were conducted under aseptic conditions. To avoid knee effusion, arthrocentesis was performed prior to the intra-articular HA injection. A physician experienced in musculoskeletal injections performed the procedure; the same technique was used for all patients.
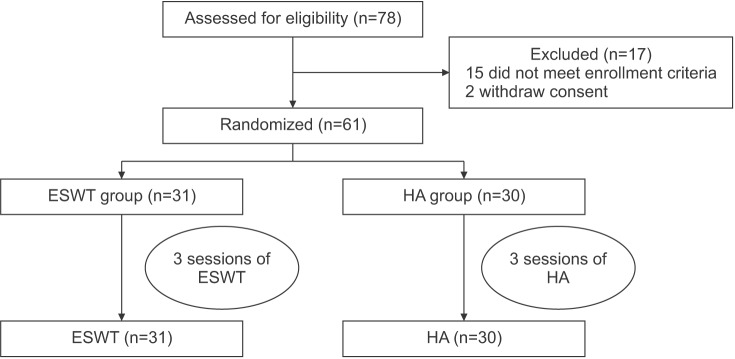
A flow diagram, showing the treatment process and assessment. ESWT, extracorporeal shock wave therapy; HA, hyaluronic acid.
No patients were allowed to receive additional treatments such as physical therapy, acupuncture, steroid injection, and analgesics. They were asked to avoid weight bearing motions that imposed an excessive burden on the affected knee, such as standing for a long time, jogging, and lifting heavy objects for the first 48 hours after each treatment. Low-level physical activities were recommended.
Outcome measures
The primary outcome measure was pain intensity as indicated by the visual analogue scale (VAS). The scores were based on pain intensity, where 0 and 10 points represent an absence of pain and a maximal intensity level of pain, respectively [20]. The VAS in this study measured the average pain intensity that patients suffered during their normal lives. The secondary outcome measurement was disability. In this study, the physical functions were evaluated using both patient-reported measures and performance-based measures. The Western Ontario and McMaster Universities arthritis index (WOMAC) and Lequesne index were applied as patient-reported measures, and the 40-m fast-paced walk test and 9-step stair-climb test (SCT) were employed as performance-based tests.
The WOMAC, a validated disease-specific self-reporting questionnaire, is an examination for evaluating OA symptoms [21]. The index is a 24-item questionnaire, grouped into three subscales including pain, stiffness, and physical function. The total score is calculated by adding the points of the three subscales. The WOMAC scores range from 0 (best) to 96 (worst) points which are based upon the severity of symptoms. The Lequesne index includes 11 questions regarding knee discomfort, endurance of ambulation, and difficulties in daily life. It is scored as follows: 24 points as the maximum score represents the heaviest dysfunction; 1–3 points indicate a mild dysfunction; and 7 points or less denote acceptable functional status [22]. The 40-m fast-paced walk test measures short-distance walking activities. The patients were asked to walk at the fastest speed as safely as possible, but not to run. They walked a total of 40 m by repeating a 10-m walk departing at a starting point and then walking another 10 m by returning to the starting point. The score was the time required to complete this task [23]. The 9-step SCT is an examination for measuring the time required to go up and down 9 stairs (stair height 20 cm), and it was measured on a centisecond basis with a stopwatch. The patients were required to go up and down the stairs safely and comfortably at their normal speed. If necessary, they could use walking aids, which was duly recorded [24].
Targeting all patients, the VAS, WOMAC, Lequesne index, 40-m fast-paced walk test, and SCT were measured before treatment, and at 1 month and 3 months after the last treatment session. All of the outcome measurements were performed by physicians not involved in any randomization and treatment protocols.
Statistical analysis
A Student t-test was conducted to compare the homogeneity of the baseline variables between the two treatment groups (ESWT group and HA group). The treatment effect over time within each group and the difference in the treatment effect between the two groups were evaluated using repeated measures of ANOVA. All statistical analyses were conducted using the Statistical Package for Social Sciences (SPSS) ver. 22.0 (IBM, Armonk, NY, USA). Data were presented as mean±standard deviation. A p-value less than 0.05 was considered statistically significant.
RESULTS
Patient characteristics
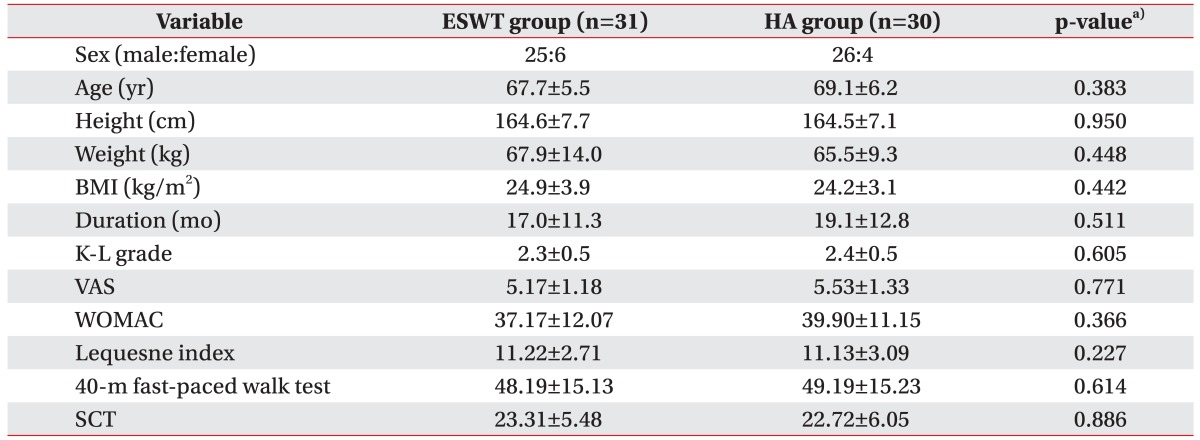
No statistical differences in sex, age, height, weight, body mass index, disease duration, K-L grade, VAS score, WOMAC score, Lequesne index score, 40-meter fast-paced walk test score, SCT score were found in the two groups before treatment (Table 1). Although some patients complained of weak side-effects in the treated regions, no treatment was suspended due to serious side-effects or complications.
Pain intensity
At baseline, the VAS scores were 5.17±1.18 in the ESWT group and 5.53±1.33 in the HA group. A month after treatment, the VAS scores were down by 3.33±1.42 in the ESWT group and by 2.93±1.48 in the HA group; at 3 months post treatment, the scores were further reduced to 2.93±2.08 in the ESWT group and 2.63±2.09 in the HA group. Compared to the baseline, the VAS scores decreased significantly up to the 3-month follow-up in a time-dependent manner in both the groups (p<0.01 for time effect). No significant differences were observed between the two groups at 1 month and 3 months after treatment (p>0.05) (Fig. 2).
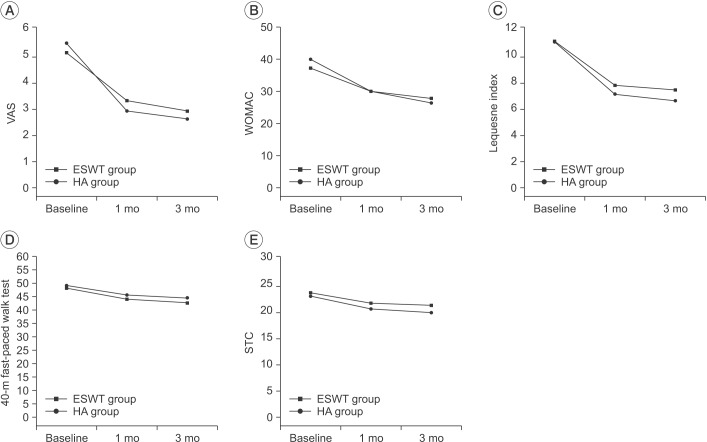
The visual analogue scale (VAS) score (A), Western Ontario and McMaster Universities arthritis index (WOMAC) score (B), Lequesne index score (C), 40-m fast-paced walk test score (D), and stair climb test (SCT) score (E) at baseline and 1- and 3-month follow-ups after treatment in the ESWT group and the HA group. These figures show significant improvement for both groups in all treatment outcomes for the entire period (p<0.01 for time effect, p>0.05 for group-time interaction).
Physical function
The baseline scores for WOMAC were 37.17±12.07 in the ESWT group and 39.90±11.15 in the HA group. At 1 month post treatment, the WOMAC scores were 30.03±10.04 and 29.97±8.11, respectively; at 3 months post treatment, the scores had further reduced to 27.73±10.13 and 26.37±8.00, respectively. The Lequesne index was recorded as 11.22±2.71 and 11.13±3.09 in the ESWT group and the HA group, respectively, at baseline. At 1 month post treatment, the scores were 7.83±2.40 and 7.15±2.40, respectively; at 3 months post treatment, the scores were 7.47±2.40 and 6.65±2.65, respectively. The 40-m fast-paced walk test baseline scores were 48.19±15.13 in the ESWT and 49.19±15.23 in the HA group. After 1 month, the scores were 44.06±14.20 and 44.59±15.00, respectively; at 3 months after treatment, the scores were 42.70±14.09 and 44.49±14.31, respectively. The baseline SCT scores were 23.31±5.48 in the ESWT group and 22.72±6.05 in the HA group. At 1 month post treatment, the scores were 21.31±5.34 and 20.26±4.49, respectively; at 3 months post treatment, the scores were 20.94±5.22 and 19.57±4.12, respectively.
All of the evaluated test scores decreased significantly in both groups up to the 3-month follow-up in a time-dependent manner compared to baseline levels (p<0.01 for time effect). No significant differences were observed between the groups at 1 month and 3 months after treatment for all tests (p>0.05) (Fig. 2).
DISCUSSION
Non-operative treatments are administered to many patients with knee OA to alleviate the symptoms and treat functional limits [25]. A combined treatment of non-pharmacological and pharmacological therapies was recommended as an ideal knee OA treatment protocol in the 2012 American College of Rheumatology guidelines [26]. ESWT has been widely used for treating a variety of musculoskeletal diseases such as calcifying shoulder tendinitis, lateral epicondylitis of the elbow, and plantar fasciitis [6]. It has just recently been proposed as a treatment for knee OA treatment. OA is characterized by pathological changes of the articular cartilage and subchondral bone [1]. The exact ESWT mechanism for joint OA is not clear but some studies have demonstrated the effects of ESWT on OA pathogenesis. Some animal studies have also reported the effects of ESWT on the articular cartilage and subchondral bone. Wang et al. [111227] proved the effects of ESWT on knee OA through pathohistological and immunohistochemical analysis. They reported that chondroprotective effects were observed in the impaired cartilage and OA-induced changes were reinstated by applying ESWT to the osteoarthritic knees of rats. Zhao et al. [9] demonstrated that nitric oxide (NO) formation was reduced in the synovial cavity of the knee joint, and chondrocyte apoptosis was also inhibited by applying ESWT to rabbits with OA. This approach resulted in a decrease in the catabolic rate within the osteoarthritic joint and induced disease-modifying effects.
In this study, ESWT as well as intra-articular HA injections were effective in ameliorating pain and functional disability (p>0.05). The curative effects of ESWT on knee OA corresponded to the results found in previous studies. Zhao et al. [13] reported pain reduction and improved knee functions in patients with knee OA in comparison to a placebo at 12-week follow-up after ESWT. Kim et al. [14] also reported that ESWT was effective in alleviating the knee pain of OA patients and improving their physical functions, and that both medium-energy ESWT (0.093 mJ/mm2) and low-energy ESWT (0.040 mJ/mm2) were effective. However, greater curative effects were shown in medium-energy ESWT group when comparing the two groups. Cho et al. [15] conducted ESWT on chronic stroke patients with knee OA. They revealed that ESWT was effective in both pain reduction and physical function improvement and that Doppler activity increased when observed with ultrasonic waves. ESWT has several advantages. Specifically, it is noninvasive, convenient, does not require hospitalization and has fewer complications. ESWT is a good alternative to avoid such risks associated with intra-articular HA injections such as bleeding, post-injection soreness, infection, allergy, and needle phobia.
A recent study recommended intra-articular HA injections for knee OA [4]. It has been widely reported and proved that intra-articular HA injection is relatively safe and effective in reducing pain and ameliorating physical functions [25]. According to an updated meta-analysis study, intra-articular HA injections showed a significant effect on pain relief and dysfunction improvement at 3 months after knee OA treatment [28]. The exact mechanism for HA has not been elucidated; however, a proposed mechanism for the HA effect was reported. Specifically, HA showed chondroprotective effects by binding to a cluster of differentiation 44 (CD44) receptors, reduced chondrocyte apoptosis, indicated anti-inflammatory effects by affecting interleukins and leukocytes, facilitated the synthesis of proteoglycan and glycosaminoglycan, showed physical effects like joint lubrication and shock absorption, and had a painkilling effect [29].
We conducted performance-based tests to supplement the patient-reported measures. In this study, performance-based measures improved in comparison with the baseline levels (p<0.01). Such improved performance-based measures in the HA group correspond to the results of prior studies. Petrella et al. [30] administered intra-articular hyaluronate sodium to knee OA patients and proved its effects by measuring their physical functions with VAS, WOMAC, self-paced walking, and stepping test. Sun et al. [31] demonstrated the effects of intra-articular HA on pain, physical function, and balance in patients with knee OA. Marks [32] analyzed factors affecting stair walking in female knee OA patients and found that pain was more strongly correlated with stair-walking time than with age, weight, height, and disease severity. Considering such study results, we conclude that the improved performance-based functions detected in the ESWT and the HA groups are associated with pain reduction after treatment.
This study has some limitations. Placebo effects were not evaluated since no control groups were included in the study. The sample size was relatively small and the follow-up period was short. Factors relevant to ESWT effects (such as energy intensity and treatment intervals) were not investigated. Thus, future studies are required that include control groups, a large sample size, long-term follow-up, and evaluation of the contributing factors affecting the treatment effects.
To conclude, the ESWT and intra-articular HA therapy are effective in alleviating pain and improving physical functions in patients with knee OA. Statistically, no significant differences in curative effects were observed between the two treatments. Therefore, ESWT can be considered another option that is as effective as intra-articular HA injections for treating knee OA.
Notes
CONFLICT OF INTEREST: No potential conflict of interest relevant to this article was reported.