- Search
| Ann Rehabil Med > Volume 46(5); 2022 > Article |
|
Abstract
Objective
To determine the characteristics of pediatric spinal cord injury (SCI) in South Korea from 1990 to 2019.
Methods
This single-centered retrospective study included pediatric SCIs. Individuals were divided into the following five groups according to onset age: ≤5, 6–12, 13–14, 15–17, and 18–19 years. The severity of complete injury was graded according to the American Spinal Injury Association impairment scale A (AIS A). Incomplete injury was graded according to AIS B, C, and D. Pearson chi-square test was used for statistical analysis.
Results
Of the 267 individuals included, 216 (80.9%) had traumatic SCIs (male-to-female ratio of 3.2:1), and 51 (19.1%) had non-traumatic SCIs (male-to-female ratio of 0.7:1). In the traumatic SCI group, 192 (88.9%) individuals were ≥15 years at the time of injury (males, 78.6%). The most common etiologies of traumatic SCIs, ranging from most to least common, were accidents related to motorcycles, falls, cars, and diving. In the non-traumatic SCI group, inflammatory (33.3%) and neoplastic (25.5%) etiologies were found to be the most common ones.
Conclusion
We found that traumatic SCIs incidence in the pediatric population was high, particularly in male individuals aged 15–19 years. The non-traumatic SCIs mostly cause paraplegia and incomplete injury. Therefore, it can be used as a basic data for the evaluation, treatment and prevention strategy of pediatric patients with SCI.
Spinal cord injury (SCI) involves damage to the spinal cord that causes temporary or permanent changes in its function. SCIs significantly alter every aspect of a person’s life, usually irreversibly [1–3]. Pediatric SCI (PedSCI) is further complicated by issues related to ongoing physical and emotional growth. After PedSCI, there is approximately a 50%–83% reduction in life expectancy [4]. In addition, given the early occurrence, individuals with PedSCI require substantially long-term care than adults with SCIs [5]. PedSCI have a large proportional composition of cervical SCI than an adult due to different vertebral configuration, incomplete ossification, relatively larger head size, and more ligamentous laxity [6]. Moreover, the socioeconomic burden of PedSCIs is greater when considering disability-adjusted life years as an endpoint [7].
PedSCI is rare compared to adults and the incidence rate of PedSCI has been studied at 0.9–27 per million per year in Europe and 14.8–20 cases per million per year in the United States [5]. A low incidence and prevalence may result in varied experiences in treatment and a low priority for the development of related research and care programs. In the United States, annual data on SCIs are systematically published by the National Spinal Injury Statistical Center (NSCISC) [8]. However, there are few national spinal injury statistics on PedSCIs in South Korea. There are few previous studies that focused on the PedSCI [9]. Therefore, this study aimed to investigate the characteristics of PedSCI in South Korea, along with its etiology and related demographic characteristics of affected individuals, and to identify prevention strategies to reduce the incidence of PedSCI.
The Ethics Committee of the Institutional Review Board of the National Rehabilitation Center (NRC) in South Korea approved this study (No. NRC-2019-04-025). The requirement for informed consent was waived owing to the study’s retrospective design.
Pediatric individuals with SCIs aged <20 years admitted to the NRC from 1990 to 2019 were enrolled. A total of 4,951 who were hospitalized with SCI were identified. Of these, 267 pediatric individuals were enrolled in this study after excluding the following: those aged ≥20 years at the time of onset (n=4,076), those who were re-admitted (n=318), those with onset dates before 1990 (n=149), and those with unknown onset date or incomplete data (n=141).
Individual data on sex, admission data, age of onset, cause of injury (injury etiology), American Spinal Injury Association impairment scale (AIS) score [10], and neurological information on the degree of impairment were collected. The neurological level of injury and the degree of impairment after the injury were assessed by a physician. In most cases, examinations had been performed during the first week after admission. The etiologies of traumatic SCI (TSCI) and non-traumatic SCI (NTSCI) were classified according to international SCI datasets [11,12]. TSCIs were classified as those caused by land transport accidents, falls, violence, sports, and others, including iatrogenic causes and in those cases where objects fell on the spine. NTSCIs were classified as those caused by neoplasm, inflammation, infection, degeneration, vascular issues, and others including syringomyelia and meningomyelocele. To identify age-related differences, the individuals were classified into the following age groups according to the International Spinal Cord Injury Data Set [11]: ≤5 years, 6–12 years, 13–14 years, 15–17 years, and 18–19 years.
All statistical analyses were performed using IBM SPSS Statistics for Windows, version 26 (IBM Corp., Armonk, NY, USA). Pearson chi-square test was used to compare categorical data, and statistical significance was set at p<0.05.
According to the cause of injury, 216 individuals had TSCIs and 51 had NTSCIs. The mean±standard deviation onset age of SCI in all study individuals was 15.7±4.5 years. Most individuals were 15–19 years old (79.8%). The most common age at injury of TSCI and NTSCI was 19 years (n=64) and 14 years (n=9), respectively. There was a significantly higher male-to-female ratio in the TSCI group (χ2=10.603; p<0.05) (Table 1).
Among the patients with traumatic spinal cord injury patients admitted to the NRC during the 1990–2019 period, 6.3% were aged <20 years. The mean age of the individuals with TSCI was 16.4±3.8 years. Of the 216 individuals with TSCI, 192 (88.9%) were ≥15 years old (male, 78.6%; n=151) (Tables 1, 2). And of 216 patients with TSCI, 10 patients (4.6%) were accompanied by hemorrhagic stroke.
Among TSCIs, motor vehicle-related complete SCI was observed in 60.2% of individuals, and motor vehicle-related incomplete SCI was observed in 39.8%. In the case of TSCI, the proportion of patients with complete injury was high. The most common level and severity of injury were complete paraplegia (n=78; 36.1%), followed by complete tetraplegia (n=52; 24.1%) (Table 2).
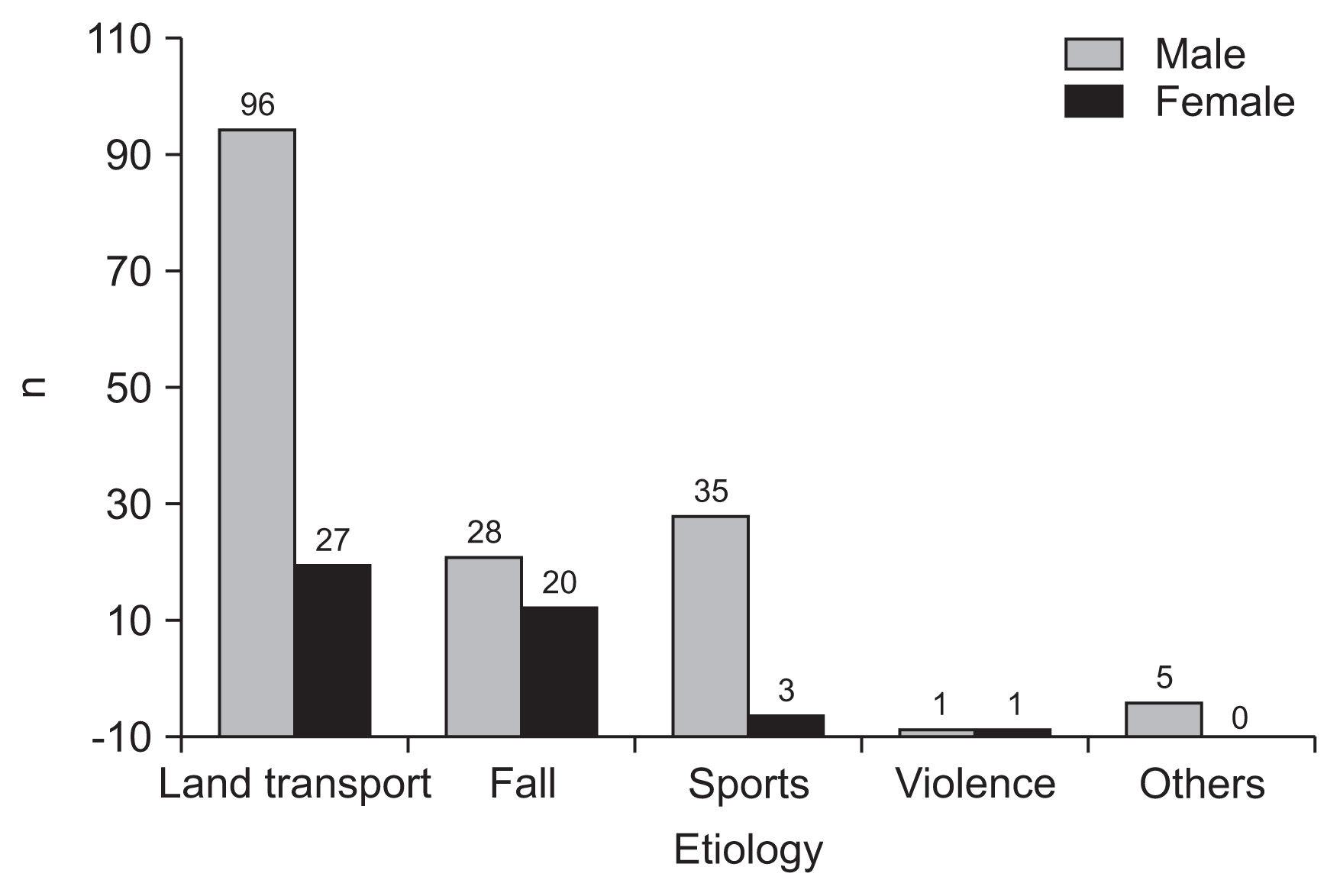
The most common cause of TSCIs was land transport accidents (n=123; 56.9%), followed by falls (n=48; 22.2%) and sports-related injuries (n=38; 17.6%) (Figs. 1, 2 and Table 2). Land transport accidents were the most common cause in both male and female participants. The second most common cause in male participants was sports, whereas in female participants it was falls (Fig. 2). There was no statistical significance between etiology and severity of injury. (p>0.05)
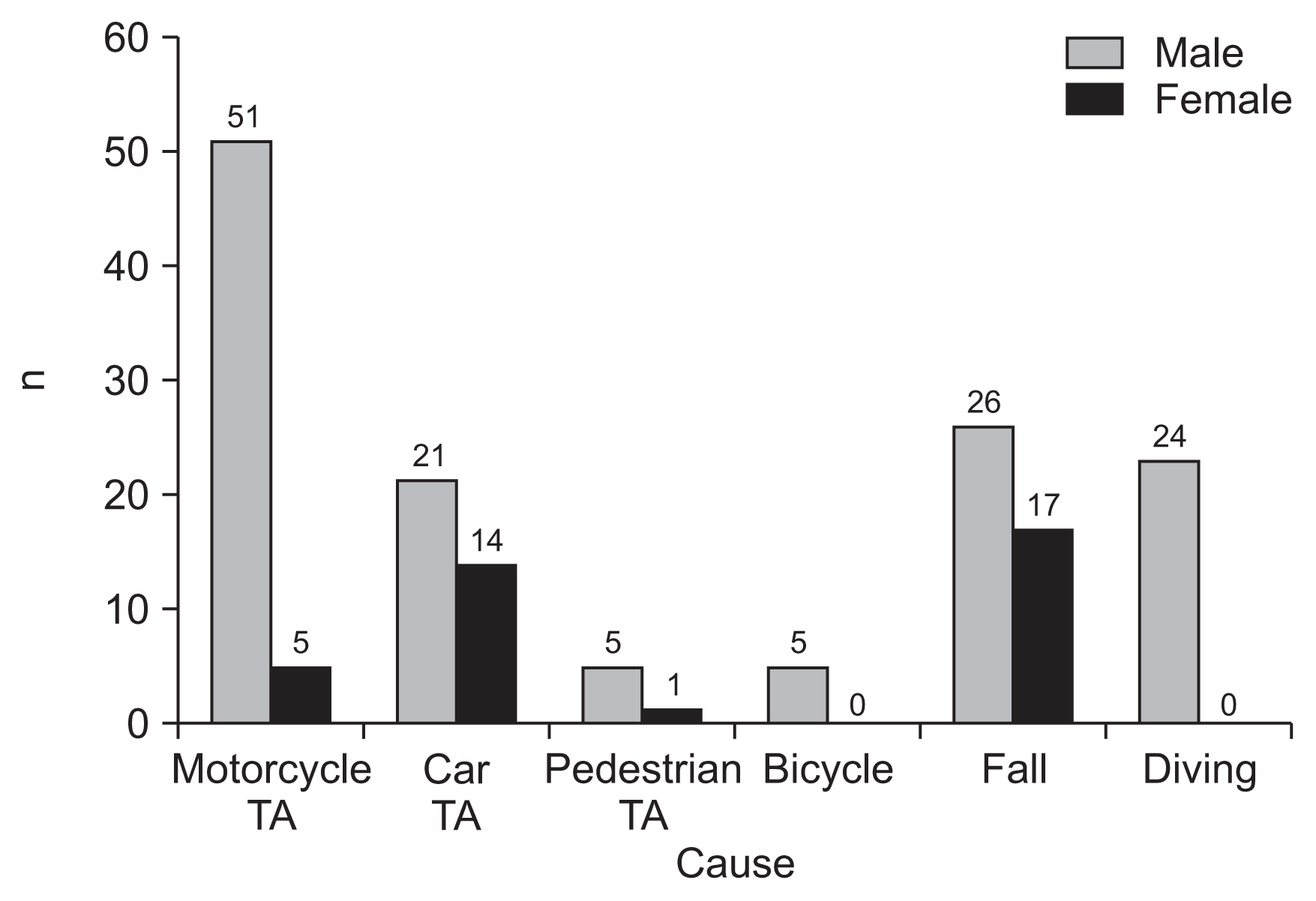
The causes of TSCI in the aged ≥15 years, were analyzed in detail (Fig. 3). The male-to-female ratio was 3.6:1. Land transport accidents (n=106; 55.2%) were the most common cause of TSCIs, followed by falls (n=43; 22.4%) and sports-related injuries (n=37; 19.3%). Of the land transport accident cases, 56 were due to motorcycle accidents, 35 were due to in-car traffic accidents, and six were due to pedestrian traffic accidents. Diving (n=24) was the most common cause of sports-related injuries (Fig. 3). According to sex, the most common cause of TSCIs in male participants was motorcycle accidents (n=51), followed by falls (n=26) and diving (n=24). In female participants, the most common cause of TSCIs was falls (n=17), followed by car traffic accidents (n=14) and motorcycle accidents (n=5) (Fig. 3).
According to the time of injury, TSCI occurred the most in June (n=26), July (n=27), and August (n=28) and the number of patients was below the average (n=18) in all the other months. There was no statistical significance between the month of the accident and the causes of TSCI (χ2=54.269; p>0.05). During the study period, the month with the most pediatric TSCIs was August (n=28), and the least was April (n=9).
The mean age of participants with NTSCI was 12.5±5.4 years. Of the 51 with NTSCIs, 12 (23.5%) had tetraplegia, 39 (76.5%) with paraplegia, 15 (29.4%) with complete injury, and 36 (70.6%) had incomplete injury. The most common injury level and severity were incomplete paraplegia (n=25; 49.0%), followed by complete paraplegia (n=14; 27.4%), incomplete tetraplegia (n=11; 21.6%), and complete tetraplegia (n=1; 2.0%) (Table 3).
Regarding etiology, inflammatory causes (n=17; 33.3%) were the most common, followed by neoplasm (n=13; 25.5%), vascular (n=8; 15.7%), infection (n=5; 9.8%), and other causes (n=5; 9.8%). There was no difference in etiology according to age.
In total, 97 individuals were hospitalized from 1990–1999, 112 from 2000–2009, and 58 from 2010–2019. The proportion of individuals with TSCI according to the year of onset was 87.6% (n=85) in 1990–1999, 77.7% (n=87) in 2000–2009, and 75.9% (n=44) in 2010–2019, with no difference between those with TSCI and NTSCI in terms of the year of onset. In addition, the severity of injury and site of injury did not differ in terms of the year of onset.
This study was conducted to describe the characteristics of PedSCI in Korea. Although PedSCI is a catastrophic event, it is difficult to study because it is relatively rare [5], and there are few prior research data on PedSCI in Korea. According to the results of our study the incidence of TSCIs in the PedSCI population in South Korea is high, particularly in males aged ≥15 years. The most frequent causes of TSCIs, in aged ≥15 years, were motorcycle accidents, followed by falls, car accidents, and diving. The causes of inflammatory and neoplasm accounted for more than half of patients of NTSCI. In addition, there were more patients with paraplegia and incomplete injury than those with tetraplegia and complete injury, respectively.
Vogel et al. [13] reported that 3%–5% of TSCIs occur in individuals aged <15 years and 15%–20% in <20 years old; however, these rates vary depending on the country and institution investigated [14,15]. In this study, 6.3% of TSCI patients admitted to the NRC during the 1990–2019 period was aged <20 years.
Pediatric patients with TSCI had a mean age of 16.4±3.8 years, and 88.9% with trauma were aged 15–19 years. This is similar to that in the 2021 United States NSCISC annual report, where individuals with TSCI aged 15–19 years accounted for 90.4% of all those <20 years old [16]. Compared with those aged <15 years, the number of cases due to land transport accidents, falls, and sports-related injuries increased rapidly in participants aged ≥15 years. In those ≥15 years old, land transport accidents were the most common cause of PedSCI, with motorcycle injuries accounting for the majority. Thus, reducing motorcycle accidents in ≥15-year-old boys would appear to be an essential injury prevention strategy.
Falls mainly involved accidents and suicide attempts [17]. Particularly in ≥15-year-old females, falls were the most common cause of SCIs, including suicide attempts. In a systematic review, Kennedy and Garmon-Jones [18] reported that approximately 6.8% of SCIs are due to suicide. According to the 2020 Korean Youth Risk Behavior Survey of the Korean Ministry of Health and Welfare, approximately 2% of middle- and high-school students in South Korea attempted suicide within the previous 12 months, with females having made more attempts than males [19]. However, this retrospective study relied on medical records; thus, whether the cause of the fall was an accident or a suicide attempt was not clearly noted, making it difficult to determine the exact number of falls attributable to suicide attempts.
All diving accidents involved males and mainly occurred in small outdoor pools and ravines in tourist locations. Diving accidents can lead to quadriplegia due to cervical SCI, and in this study, 24 individuals with diving-related TSCI admitted to the hospital were tetraplegic, of which 75% were motor-complete SCIs. The 58.3% in diving injuries occurred in July (n=6) and August (n=8). In the United States, violence-related SCIs have been reported to comprise the third most common SCIs among those aged <15 years, and the second-highest among those aged 15–30 years [16]. However, in countries with restrictions on firearm use, such as Ireland, SCI cases due to violence are rare [20]. In this study, SCI due to violence was noted in 2 cases (0.9%). Thus, to prevent the occurrence of TSCIs in South Korea, it is necessary to discourage dangerous behaviors in middle- and high-school students aged ≥15 years, particularly among male students. The prevention of motorcycle and diving accidents should also be emphasized.
Regarding NTSCIs, inflammation (n=7) was the most common cause in those aged 0–9 years, with six cases of these being due to transverse myelitis. In those aged 10–19 years, neoplasm (n=13) was the most common cause. In pediatric NTSCI studies conducted in the United Kingdom Ireland, and Australia, transverse myelitis and neoplasm were the major causes of pediatric NTSCIs [20–22]. Meningomyelocele was a common congenital cause. As the importance of folic acid supplementation in pregnant women has become widely known since the 1990s, the incidence of spina bifida has decreased [23], as reflected in our results. There were three individuals with NTSCI in the 1990s due to meningomyelocele and only one was similarly affected since 2000. Despite being a developed country, tuberculosis is still common in South Korea. In this study, there were three NTSCI cases in the 2000s caused by tuberculosis spondylitis.
The male-to-female ratio was 3.2:1 in pediatric individuals with TSCI and 0.7:1 in pediatric individuals with NTSCI. Traumatic causes suggest that males tend to be more active and engage in riskier activities than females, resulting in a lower incidence of TSCIs [5]. This study also showed that motorcycle and diving accidents with a high risk of trauma mostly occurred in males (93.7%) aged 15–19 years.
This study had some limitations. First, it was a single-centered study; thus, our results cannot represent all individuals with PedSCI in South Korea. Second, data from individuals who required intensive care, such as those needed ventilator support and those who expired in the acute phase immediately after SCI, were not included because there is no intensive care unit ward in the NRC. Therefore, multicenter, large population-based studies should be conducted to acquire a more accurate picture of PedSCI epidemiology. Finally, it was impossible to determine whether TSCIs were caused by a fall accident owing to carelessness or a suicide attempt. Hence, the proportion of falls caused by suicide attempts remains unknown.
Nevertheless, this is the largest and most comprehensive study on PedSCIs in South Korea, involving data from the past 30 years. This study also analyzed the most frequent causes of TSCIs in detail. Therefore, this study is important because this can be used as reference in understanding the characteristics of PedSCI in South Korea.
In conclusion, this study describes the characteristics of PedSCI in South Korea. The proportion of men in TSCI was high, and the number of TSCI patients increased rapidly, after the age of 15 years. NTSCI mostly caused paraplegia and incomplete injury, and inflammatory and neoplasm induced SCIs were the most common.
In this study, we identified the current status and causes of PedSCIs in South Korea. Our results can be used as reference for appropriate evaluation, therapeutic approach and prevention strategy by age and causes.
ACKNOWLEDGMENTS
This research was supported by the Korea National Rehabilitation Center (No. 21-H-01).
AUTHOR CONTRIBUTION
Conceptualization: Lee BS, Kim O. Methodology: Lee BS, Kim O. Formal analysis: Kim W, Yun H, Han SR. Project administration: Lee BS, Kim O. Visualization: Yun H, Han SR. Writing - original draft: Kim W. Writing - review and editing: Lee BS, Kim O, Kim W. Approval of final manuscript: all authors.
Fig. 1
Etiology distribution of individuals with traumatic pediatric spinal cord injury stratified by age.

Table 1
Demographic characteristics of pediatric patients with spinal cord injury according to age groups (n=267)
Table 2
Injury profiles of pediatric patients with traumatic spinal cord injuries
Table 3
Injury profiles of pediatric patients with non-traumatic spinal cord injuries
REFERENCES
1. Vitale MG, Goss JM, Matsumoto H, Roye DP Jr. Epidemiology of pediatric spinal cord injury in the United States: years 1997 and 2000. J Pediatr Orthop 2006;26:745-9.

2. Flett PJ. The rehabilitation of children with spinal cord injury. J Paediatr Child Health 1992;28:141-6.


3. Massagli TL, Jaffe KM. Pediatric spinal cord injury: treatment and outcome. Pediatrician 1990;17:244-54.

4. Shavelle RM, Devivo MJ, Paculdo DR, Vogel LC, Strauss DJ. Long-term survival after childhood spinal cord injury. J Spinal Cord Med 2007;30(Suppl 1): S48-54.



5. Chien LC, Wu JC, Chen YC, Liu L, Huang WC, Chen TJ, et al. Age, sex, and socio-economic status affect the incidence of pediatric spinal cord injury: an eleven-year national cohort study. PLoS One 2012;7:e39264.



6. Wu JC, Chen YC, Liu L, Chen TJ, Huang WC, Cheng H, et al. Effects of age, gender, and socio-economic status on the incidence of spinal cord injury: an assessment using the eleven-year comprehensive nationwide database of Taiwan. J Neurotrauma 2012;29:889-97.


7. Hall OT, McGrath RP, Peterson MD, Chadd EH, DeVivo MJ, Heinemann AW, et al. The burden of traumatic spinal cord injury in the United States: disability-adjusted life years. Arch Phys Med Rehabil 2019;100:95-100.


8. Parent S, Mac-Thiong JM, Roy-Beaudry M, Sosa JF, Labelle H. Spinal cord injury in the pediatric population: a systematic review of the literature. J Neurotrauma 2011;28:1515-24.



9. Shin H. Etiology and epidemiology of spinal cord injury in Korea. J Korean Med Assoc 2020;63:589-95.


10. DeVivo MJ, Biering-Sorensen F, New P, Chen Y. Standardization of data analysis and reporting of results from the International Spinal Cord Injury Core Data Set. Spinal Cord 2011;49:596-9.



11. Biering-Sorensen F, DeVivo MJ, Charlifue S, Chen Y, New PW, Noonan V, et al. International Spinal Cord Injury Core Data Set (version 2.0)-including standardization of reporting. Spinal Cord 2017;55:759-64.



12. New PW, Marshall R. International Spinal Cord Injury Data Sets for non-traumatic spinal cord injury. Spinal Cord 2014;52:123-32.



13. Vogel LC, Betz RR, Mulcahey MJ, Zebracki K. Spinal cord injuries and disorders in children and adolescents. In: Kirshblum S, Lin VW, editors. Spinal cord medicine. 3rd ed. New York, NY: Springer; 2019. p.926-54.

14. Chen Y, DeVivo MJ. Epidemiology. In: Vogel LC, Zebracki K, Betz RR, Mulcahey MJ, editors. Spinal cord injury in the child and young adult. London, UK: Mac Keith Press; 2014. p.15-27.
15. Saunders LL, Selassie A, Cao Y, Zebracki K, Vogel LC. Epidemiology of pediatric traumatic spinal cord injury in a population-based cohort, 1998–2012. Top Spinal Cord Inj Rehabil 2015;21:325-32.



16. National Spinal Cord Injury Statistical Center. 2021;Annual Report Complete Public Version
https://www.nscisc.uab.edu/PublicDocuments/AR2021_public%20version.pdf
.
17. January AM, Zebracki K, Chlan KM, Vogel LC. Mental health and risk of secondary medical complications in adults with pediatric-onset spinal cord injury. Top Spinal Cord Inj Rehabil 2014;20:1-12.



18. Kennedy P, Garmon-Jones L. Self-harm and suicide before and after spinal cord injury: a systematic review. Spinal Cord 2017;55:2-7.



19. Korean Statistical Information Service. Adolescent health behavior survey 2020 [Internet] Daejeon, Korea, Korean Statistical Information Service. 2020;[cited 2022 Oct 1]. Available from: https://kosis.kr/statHtml/statHtml.do?orgId=177&tblId=DT_117_12_Y071&checkFlag=N
.
20. Smith E, Finn S, Fitzpatrick P. Epidemiology of pediatric traumatic and acquired nontraumatic spinal cord injury in Ireland. Top Spinal Cord Inj Rehabil 2017;23:279-84.



21. Galvin J, Scheinberg A, New PW. A retrospective case series of pediatric spinal cord injury and disease in Victoria, Australia. Spine (Phila Pa 1976) 2013;38:E878-82.